Upper-extremity support AM-AO-KG


 Upper limb brace
Upper limb brace Breathable
Breathable Class I medical device
Class I medical device Durable
Durable Innovative
Innovative Recommended by specialists
Recommended by specialists Skin-friendly
Skin-friendly Universal size
Universal sizeCLEVER
Shoulder rotation brace
Description
The shoulder rotation brace CLEVER with rotation angle regulation is an efficient immobilizer for treatment of an arm fracture, a dislocated shoulder or rotator cuff surgery in post-operative and post-traumatic conditions. The Arm rotation brace AM-AO-KG called CLEVER shall be prescribed by the doctor or professional orthopaedic specialist. According to the newest medical investigations recommends the application of positioning of shoulder in lateral rotation in the cases of treatment of shoulder dislocations.
The support sleeve is closed by the circumference stabilization strap.
Construction: The Orthosis is made of Active3D and AirDistance II.
CLEVER 2 is equipped with semi rigid, anatomically shaped forearm sleeve, with self-regulation length.
The orthosis CLEVER is provided with auto adhesive closure bands, which allow easy adjustment of their length additionally equipped with soft protectors. The construction of bands helps to obtain maximum unloading of the shoulder.
The orthosis is equipped with hip aluminum frame with shoulder abduction system. The aluminum frame is anatomically shaped, perfectly fits to the patient body improving stabilization of the joint.
The product offers possibility to fit a handle support for a hand or silicone ball for hand exercising.
Properties
– the support maintaines the limb in abducent position, special design of the allumninum splint enables fluent adjustment of the movement range in the humeral joint (from 20 internal rotation to 15 of outside rotation)
– ARC; from 15 internal rotation to 70 of outside rotation – ARC-XR
– special foam handle enables exercisse of forearm and hand muscles as well as guarantees placement of the wrist in the medium position)
– the suspension tape of the support is equipped with soft padding in the neck area which assures beter comfort of use and no pressure on the artery.
– the support can be used for left and right upper limb
– wide range of adjustment of the VELCRO tapes as well as the elasticity of the abducent splint make the support universal: one size for all
– abducent brace fulfils all the terapeutic functions leaving the area of the hummeral joint free to enble easy change of dressing or physiotherapeutic treatment without taking off the whole stabilizing construction.
Purpose of use
The shoulder rotation brace, type AM-AO-KG, can be used in case of all injuries or disorders in the area of the shoulder, requiring support or immobilization of the upper limb. It is also used after the surgery. The brace is especially useful as means of immobilization after the artroscopy or open surgery of the humeral joint. It is used to stabilize and place the humeral joint in the required position.
Indications
The orthosis is recommended to support and partially immobilization of the Upper Limb in case of Plegia or Paralysis.
– Post-operative and Post-Traumatic treatment of the shoulder required positioning of the shoulder and arm in rotation angle
– Rotator Cuff Plastic Surgery
– Anterior Luxation
– Fracture of the head of humerus
– Bankart injury of shoulder dislocation
– Post arthroscopy of shoulder
– Shoulder prosthesis
– Frozen Shoulder- Adhesive Capsulitis
ATTENTION: the rotation brace of the upper limb AM-AO-KG is not designed to be used again by another patient
Contraindications
In case of skin changes in place where the brace touches the skin (abrasions, injuries, skin eczema) use of the brace should be limited
In case of skin changes in place where the brace touches the skin it is necessary to contact your doctor.
Attention:
– Use of the brace without following this instruction may result in severe complications
– Patients using the brace without doctor’s or therapist recommendation, use the brace on their own risk
Sizes
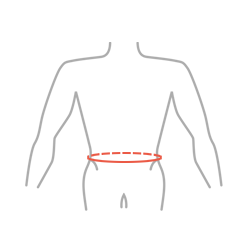
| Size | Waist circumference | How to measure |
| Single size | min 65 cm – max 145 cm (min 25,6″ – max 57,1″) |
 |
Total height of the product: 21 cm (8,3″)
Total length of product: 47 cm (18,5″)
Universal product for the right and left hand
Gallery
Technology
MATERIALS
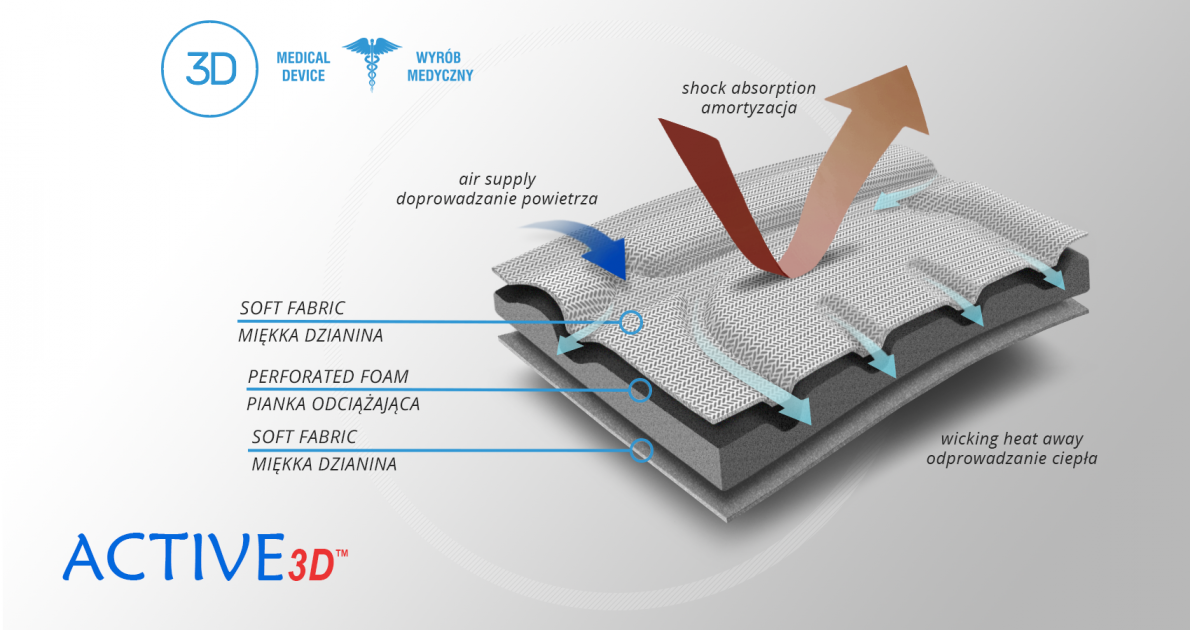
Active3D™
Active3D™ is thermoformed fabric made of special foamed, cell-closed designed material. It reduces the pressure on the body or any abrasions made by orthopaedic stays and aluminum splints. It is fully waterproof fabric and does not absorb sweat. It’s easy to clean. Due to its features, the fabric is an excellent product for making medical orthopaedic braces and orthoses. ACTIVE 3D™ has various external self-gripping layers. Our material has special, thermoformed properties and may be shaped according to the functional goals of the final braces.
AirDistance II™
AirDistance II™ is an innovative technological fabric. It’s 3-layered lamination, made of 3 parts: durable and self-gripping and polyamide layer, off-loading EVA foam and polyamide 3D spacer material. The external part is used to attach hard components. EVA foam off-loads these splints and the internal 3D spacer layer provides high air permeability and minimal water and moisture absorption. Waterproof material.
STIFFENINGS
Aluminum frame with rotating element
The hip frame is made of soft aluminum sheet connected to the arm stabilizer. The hip basket of the frame is equipped with a circumferential belt positioning the product on the patient's body, and the arm stabilizer is equipped with a splint that allows you to set the level of rotation of the upper limb in relation to the body. The frame is completely covered with a durable powder coating, neutral for the patient's skin.
PADDINGS
3D supports
3D relief supports are independent technical solutions to relieve the rigid elements of a given orthosis. These elements are made of supporting foams or EVA foam. These foams are connected with various types of skin-friendly materials and materials with an adhesive function. These pads have the appropriate shape and color adapted to the type of orthosis. They relieve both metal elements of orthoses, such as splints, stays, underwires and orthopedic drop locks, as well as other elements that should not come into direct contact with the patient's skin. These pads have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, guaranteeing the proper therapeutic effect.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.