Lower limb support COMPLEX/2R
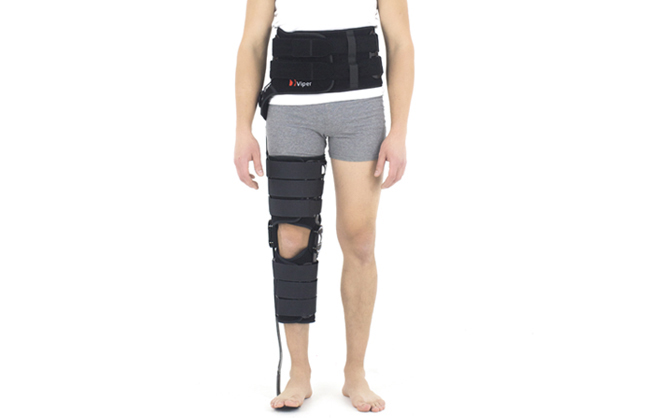

 Lower limb brace
Lower limb brace Breathable
Breathable Class I medical device
Class I medical device Dial joint 1RE
Dial joint 1RE Full range of adjustment
Full range of adjustment Innovative
Innovative Latex-free
Latex-free Polycentric joint 2R
Polycentric joint 2R Recommended by specialists
Recommended by specialists Skin-friendly
Skin-friendly Universal size
Universal sizeCOMPLEX
HKAFO ORTHOSIS WITH DYNAMIC CARBON FOOT AND HIP AND KNEE ROM ADJUSTMENT
Description
HKAFO orthosis (hip, thigh and calf part) is made of innovative fabric ACTIVE DISTANCE II as well as ACTIVE DISTANCE
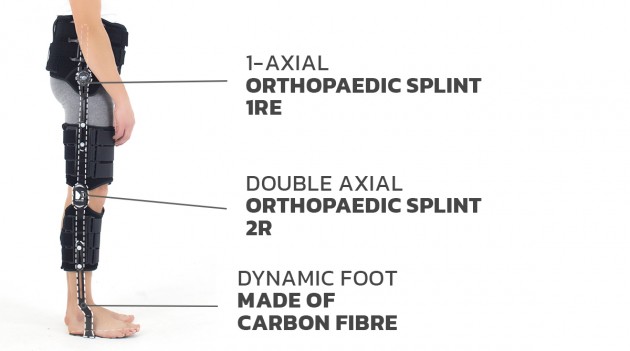
The device is equipped with orthopaedic splints and dynamic carbon foot. The splint are made of high quality aluminium hot covered by special powder surface, what makes them neutral to patient’s sweat.
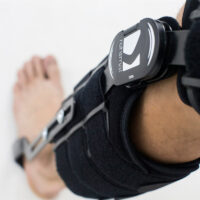
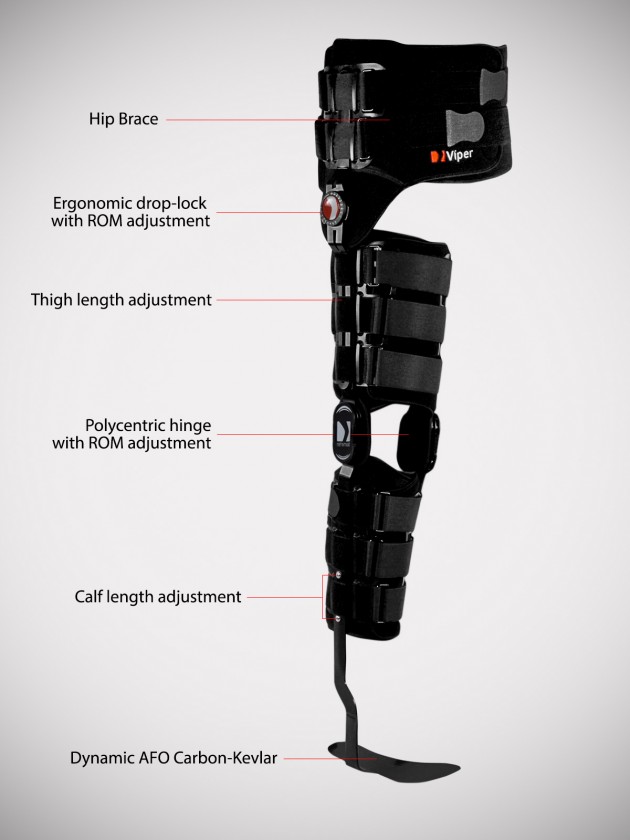
The hip joint is equipped with 1-axial hinge with ROM adjustment in every 15o and the knee joint holds 2-axial polycentric hinge with ROM adjustment in every 20o. The ankle joint is non hinged.
For your special order, to ankle joint we can add our orthopaedic hinge 1R with ROM adjustment in every 15°.
Each of these drop-locks can be adjusted in the required position and blocked against the displacement.
The thigh and calf part of orthosis provides height adjustment what allows to fit the device according to individual patient’s size.
The carbon foot is resilient in the front and heel part. Semi-rigid spiral splint and shin pelotte stabilize the orthosis on calf. Our carbon foot has 3 points of support: in the front middle-tibia, the tibial tuberosity and proximal tibial and fibula epiphyses. Because of that, all forces accumulates during limb loading and helps the patient with foot dorsal flexion. The dynamic ankle-foot orthosis (AFO) allows patient to have medial-lateral control of the foot and ankle complex due to strong lateral fixation of lower leg. The gait is improved and more aesthetic. The AFO construction is very light and easy to putting into shoe.
HKAFO orthosis is equipped with three pairs of Velcro straps: 2 straps for pelvis, 2 for thigh and 2 for calf. The thigh and calf part of device holds 2 pairs of pelottes made of plastics material where our splint are attached.
Safe construction and soft and friendly fabrics make our COMPLEX unrivalled with other devices available on the market.
Indications
Double-sided COMPLEX orthosis should be used in case of:
- flaccid paraparesis;
- myelocystocele, hydromyelomeningocele;
- spinal muscular atrophy;
- muscular dystrophy / myodystrophy with upper limbs mobility.
One-sided COMPLEX orthosis should be used in case of:
- severe knee joint instability or degeneration with foot paresis (sciatica, SM, Poliomyelitis – Heine-Medin disease);
- tetraparesis – in upright position, while gait learning;
- to immobilize in rehabilitation process after knee surgeries or diseases;
- to prevent against hip, knee or ankle muscles contractures;
- to proper hip position during walking or upright position.
The COMPLEX orhosis is innovative solution in case of partly feet amputation. In this case, there is necessary to use non-standard insole. The orthosis’s construction directs forces to upper tibia what offloads the amputated foot.
The orthosis can be used as an modular device – with patient’s recovery, the doctor can stop using the hip splint. In this case, the lower limb and ankle brace exists independently.
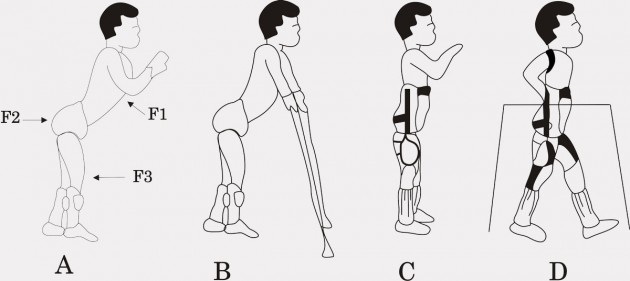
A. AFO
B. KAFO
C. HKAFO
D. HKAFO+TLSO
Sizes
TABLE 1
| Size | Waist circumference | How to measure |
|---|---|---|
| Single size | min 75 cm – max 125 cm (min 29,5″ – max 49,2″) |
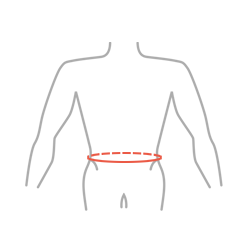 |
TABLE 2
| Size | (A) Thigh circumference 15 cm above the patella | (B) Calf circumference 15 cm below the patella | How to measure |
|---|---|---|---|
| Single size | min 40 cm – max 75 cm (min 15,7″ – max 29,5″) |
min 30 cm – max 55 cm (min 11,8″ – max 21,7″) |
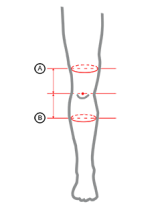 |
TABLE 3
| Size | (C) Distance between knee joint interstice and groin | (D) Distance between knee joint interstice and foot | How to measure |
|---|---|---|---|
| S | 27-37 cm (10,6″-14,6″) |
34-45 cm (13,4″-17,7″) |
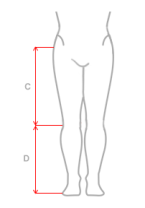 |
| M | 33-43 cm (13″-16,9″) |
40-51 cm (15,7″-20″) |
|
| L | 36-46 cm (14,2″-18,1″) |
42-54 cm (16,5-21,2″) |
|
| XL | 49-61 cm (19,3″-24″) |
TABLE 4
| Size | Foot length | How to measure |
|---|---|---|
| S | 22-24 cm (8,7″-9,3″) |
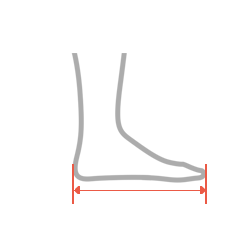 |
| M | 24-26 cm (9,4″-10″) |
|
| L | 26-28 cm (10,2″-10,8″) |
|
| XL | 28-30 cm (11″-11,6″) |
Left/right side available
Maximum user weight up to 75kg.
ANY SIZE COMBINATIONS FROM TABLES 3 AND 4 ARE POSSIBLE
Gallery
Technology
MATERIALS
ActiveDistance II™
ActiveDistance II™ is an active 3-layered fabric made of non-elastic polyamide self-gripping layer, comfort foam and cotton terry. The last one layer is responsible for maintaining your skin dry. This material is skin-friendly and has the Oeko-Tex Standard 100 certificate. Self-gripping function of ActiveDistance II™ allows to fit the braces precisely.
ActiveDistance™
ActiveDistance™ is an technical off-loading lamination with 3-layered construction. It’s made of EVA foam double-sided laminated with non-elastic polyamide layer that is easy to attach. This self-gripping function of ActiveDistance™ provides wide range of adjustment and perfect fitting. What is important, ActiveDistance™ is non-elastic fabric so it stabilizes your body in a perfect way. Thanks to using the comfort foam, the device is really soft what influences on the comfort of using it. Waterproof material.
TECHNOLOGICAL SYSTEMS
STIFFENINGS
Splint 1RE
Splints 1RE – single axis orthopedic splints, used mainly in knee joint, upper limb and hip joint braces. The characteristic quality of this splint is, apart from wide range of motion adjustment in every 15 degrees, is also hyperextension adjustment in the secured range of -15 and -30 degrees. The splints are characterized by perfect lateral joint stabilization and low weight. The set-up of the angle adjustment clock doesn’t require any tools, and special safety system prevents the change of the angle by unauthorized people. The splints are made of high quality aluminium alloy and plastic with carbon fibre. They are light and neutral to the influence of sweat and salt, properly firm, assuring correct functioning of the device. The splint and its design are patented in European Union by Reh4Mat company.

Splint 2R
Splints 2R – double axis polycentric splints, with rack gear which reflects the anatomic knee movement. Such splints are used in all knee joint braces where apart from angle adjustment lateral joint stabilization is also important. The splints are manufactured with high quality of aluminium alloy with sanded, durable cover which is neutral to the influence of sweat and salt. Required flexion and extension angle in every 20° is set by using the special pins. The splints are equipped with special oval covers to prevent the pins from falling out as well as from changing of the angle by unauthorized people. Standard length of the splints is 390 mm, but the braces with longer splints are additionally equipped with lateral stabilization of the knee joint. The splint and its design are patented in European Union by Reh4Mat Company.



PADDINGS
3D supports
3D relief supports are independent technical solutions to relieve the rigid elements of a given orthosis. These elements are made of supporting foams or EVA foam. These foams are connected with various types of skin-friendly materials and materials with an adhesive function. These pads have the appropriate shape and color adapted to the type of orthosis. They relieve both metal elements of orthoses, such as splints, stays, underwires and orthopedic drop locks, as well as other elements that should not come into direct contact with the patient's skin. These pads have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, guaranteeing the proper therapeutic effect.
Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.