Finger splint AM-SP-03

 Finger brace
Finger brace Cast replacement
Cast replacement Class I medical device
Class I medical device Innovative
Innovative Minimum device - maximum effect
Minimum device - maximum effect Universal size
Universal size Waterproof
WaterproofFINGER SPLINT
Description
Finger dislocation
Finger’s dislocation is often condition. Falling onto an outstretched finger or hitting the ball during sport training may result in dislocation of finger. It rarely go unnoticed. The finger becomes red, swollen, deformed and painful. The most common method of treatment is ice pack and finger immobilization. To do that you should use our professional finger’s splint AM-SP-03.
Product’s description
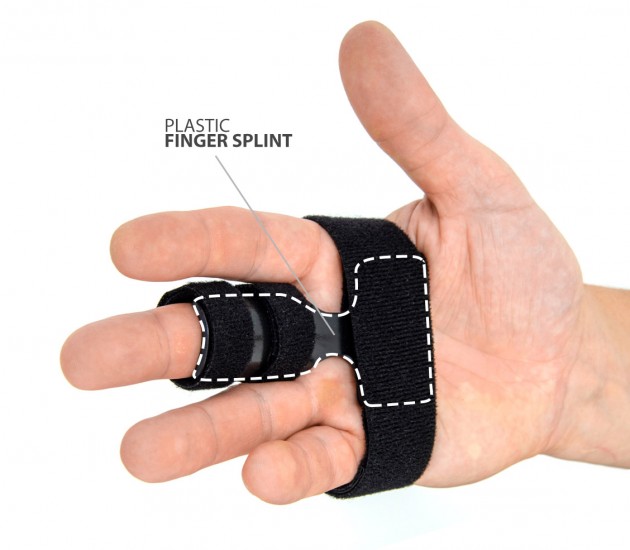
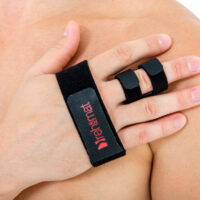
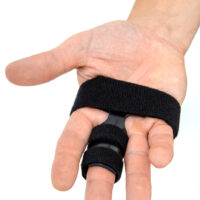
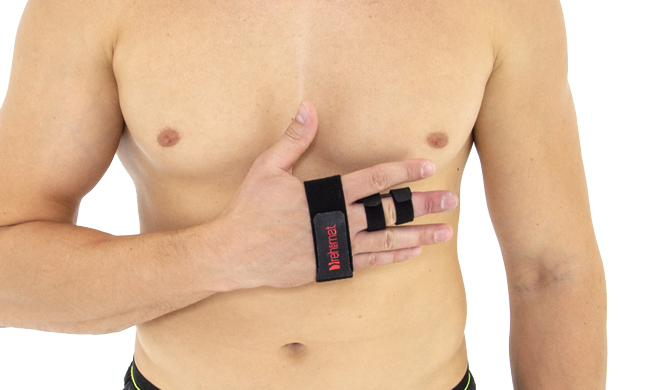
Our finger splint AM-SP-03 is a simple, innovative product used for immobilization of II-V fingers.
Made of weight light rigid plastic for perfect stabilization of phalanges. It is ergonomically anatomic shaped for excellent fitting and perfect immobilization.
Soft part of our finger’s immobilizer AM-SB-03 is made of skin friendly UniTape™.
Our finger splint AM-SP-03 is waterproof product so you can take a shower wearing it.
Our splint immobilizes the metacarpophalangeal (MCP) joints and proximal (PIP) interphalangeal joints and keeps them in neutral position in case of sprain, dislocation or fracture of the finger.
Our finger splint AM-SP-03 offers wide range of circumferential adjustment and had universal size. Also, it fits for both hands.
Our finger splint AM-SP-03 is easy to use and disinfection. It is excellent support in case of finger’s sprain, dislocation of fractures of phalanges.
Purpose of use
• II-V fingers fractures
• Injuries of collateral ligaments of MCP joints
• II-V fingers dislocations
• rheumatic diseases of II-V fingers
Sizes
| Size | Metacarpal circumference | How to measure |
| Universal | min 15 – max 25 cm min 5,9″ – max 9,8″ |
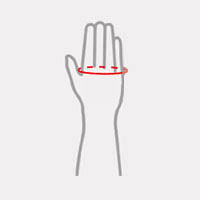 |
Fits for both hands.
Total length of the product: 23 cm (9″)
Gallery
Technology
MATERIALS
UniTape™
UniTape™ is a technological orthopedic lamination with high durability with double-sided self-gripping function. UniTape™ is very thin and easy to attach what allows to fit the braces precisely.
STIFFENINGS
Finger splint
Finger splints are the kind of simple orthoses that support the metacarpophalangeal joints (MCP). The splint is made of ABS and pre-shaped to fit to the fingers. Velcro fasteners are installed on the splint, to which the elastic circumferential bands are attached.

Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.