Lower-extremity support AM-OSK-O/1R

 Knee brace
Knee brace Breathable
Breathable Class I medical device
Class I medical device Double-sided
Double-sided Latex-free
Latex-free Unicentric joint 1R
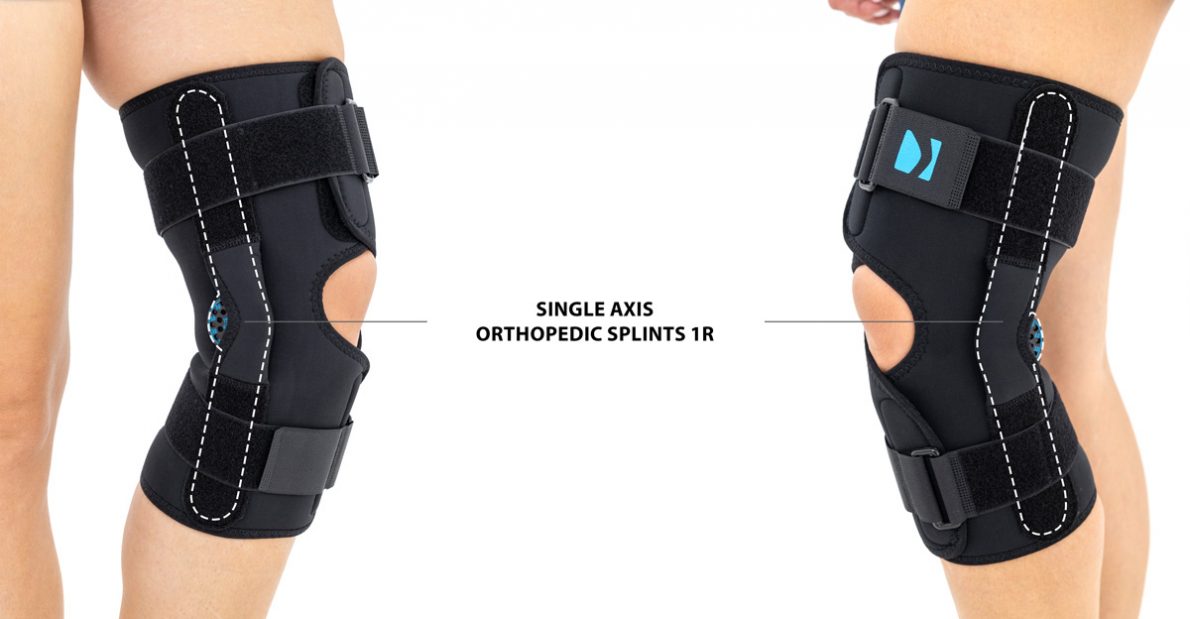
Unicentric joint 1ROPEN KNEE BRACE WITH 15° ROM ADJUSTMENT AND DROP LOCK COVER
Description
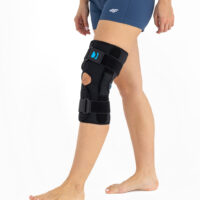
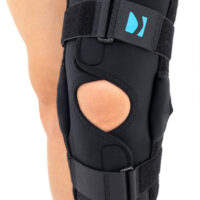
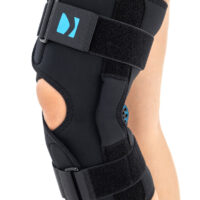
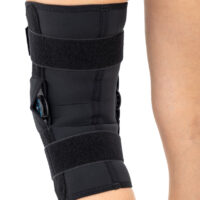
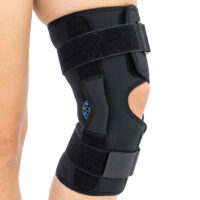
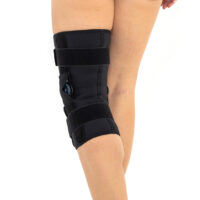
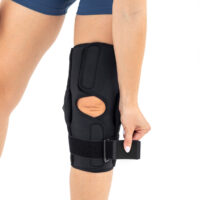
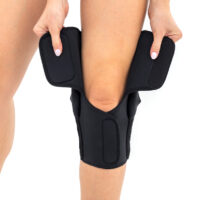
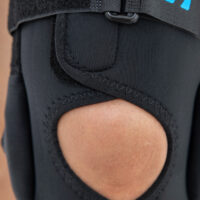
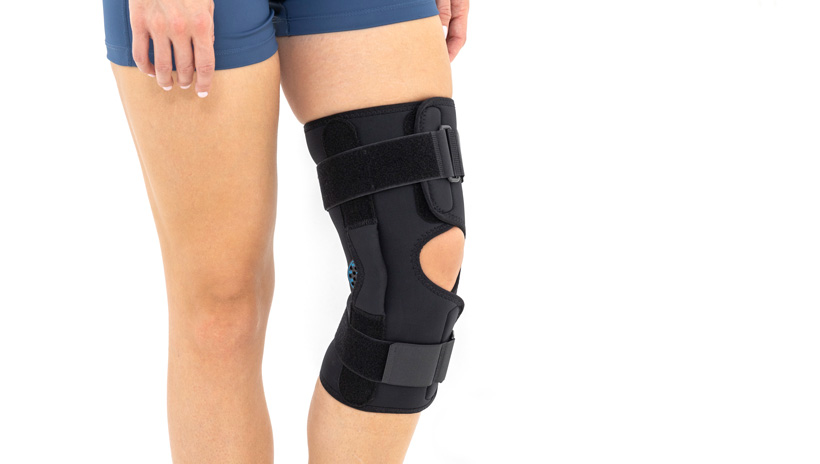
The wrap around knee brace is made of innovative ActivePren™.
It holds two independent supported sets of the Velcro tapes. The device is equipped with two side splints, which are made of carbon fibres and consists of special drop lock allowing for ROM adjustment in every 15°. this precise adjustment facilities the rehabilitation process.
The system of splints in wrap around knee brace is arranged in a shape of X letter, which allows to adhere perfectly to the limb and also enables to stabilize the joint precisely. This internal perforation allows skin to breath and reduces overheating. The wrap around construction enables to use the brace by seniors or overweight people.
Purpose of use
AM-OSK-O/1R brace should be applied in cases of:
- knee dislocation,
- knee twist and sprain,
- side instability of the knee joint,
- knee ligaments LCL, MCL and ACL injuries,
- knee ligaments reconstruction,
- other surgeries (orthopedic).
Sizes
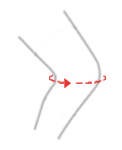
| Size | Knee circumference | How to measure |
| S | 30 – 34 cm (11,8″-13,4″) |
 |
| M | 34 – 38 cm (13,4″-15″) |
|
| L | 38 – 42 cm (15″-16,5″) |
|
| XL | 42 – 46 cm (16,5″-18,1″) |
|
| 2XL | 46 – 50 cm (18,1″-19,7″) |
|
| 3XL | 50 – 55 cm (19,7″-21,7″) |
|
| 4XL | 55 – 59 cm (21,7″-23,2″) |
Fits for both knees.
Total length of the product: 33 cm (13″)
Gallery
Technology
MATERIALS
ActivePren™
ActivePren™ is an active three-layer material consisting of two elastic jersey cover fabrics and a core made of neoprene foam. This material is characterized by softness and high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of ActivePren are the strongest and most effective stabilizing orthoses available on the market.
STIFFENINGS
Splint 1R
1R splints – single axis orthopedic splints used in all knee joint and upper limb braces, where apart from adjustment of the flection and extension angle, high lateral stabilization of the joint is not required, but important if low weight of the brace. Due to the low weight the splints are successfully used in all braced for children. They are manufactured with high quality polyamide with carbon fibres, making the splints light and neutral to the influence of sweat and salt, properly firm therefore assuring correct functioning of the device. Adjustment of the flexion and extension angle is performed with special Allen screw in every 15°. Each brace is equipped with an Allen key, which is necessary for adjustment. Standard length of the braces is: 280 and 380 mm. The splint and its design are patented in European Union by Reh4Mat Company.

Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.