AM-D-06

 Finger brace
Finger brace Cast replacement
Cast replacement Class I medical device
Class I medical device ER
ER Innovative
Innovative Orthopedics
OrthopedicsAM-D-06 – THUMB STABILIZER SPLINT AND THUMB ARTHRITIS SPICA
Description
THUMB INFLAMMATION
Inflammatory conditions of the thumb may affect the joints, tendons, and periarticular structures and are often the result of overuse, micro-trauma, or degenerative changes. They present with pain, swelling, tenderness, and limited range of motion, which makes everyday activities such as gripping, writing, or lifting objects difficult.
The use of a thumb stabilizer splint such as AM-D-06 helps limit movements that exacerbate symptoms, reduces stress on inflamed structures, and creates conditions conducive to regeneration. Thumb stabilization can be an important component of conservative treatment and rehabilitation, in accordance with a specialist’s recommendations.
Product’s description:
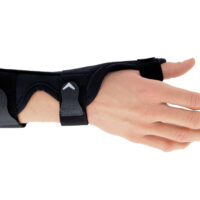
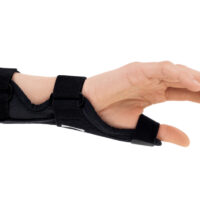
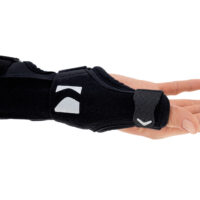
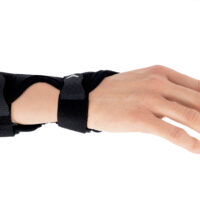
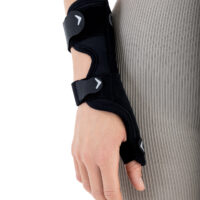
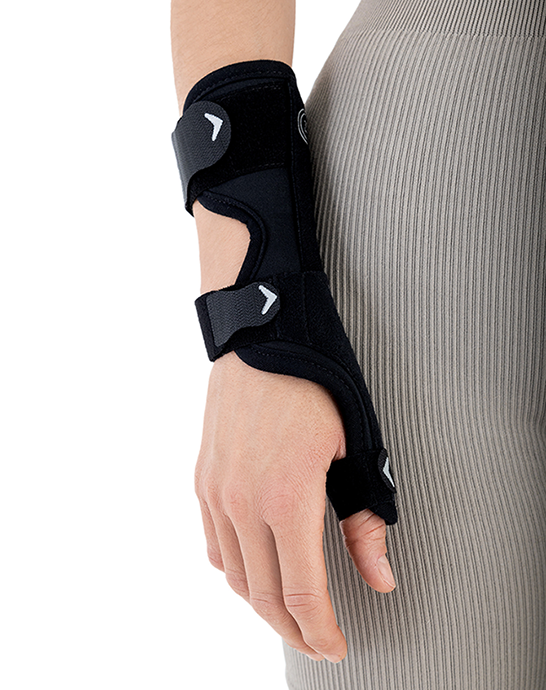
The AM-D-06 hand splint has been designed to stabilize the thumb and wrist while maintaining the greatest possible hand functionality during everyday activities. Its construction provides controlled thumb motion by limiting abduction and adduction, while also supporting the wrist joint to reduce overload and pain.
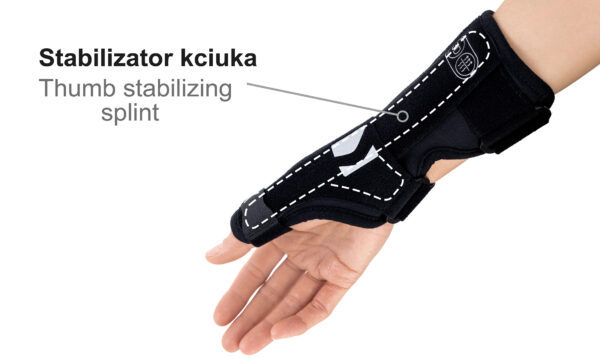
Equipped with an anatomically contoured lateral splint, it effectively stabilizes the thumb metacarpophalangeal (saddle) joint, limiting abduction movements that excessively strain the ulnar collateral ligament (UCL). Controlled immobilization reduces ligament tension, supporting healing and alleviating pain.
The AM-D-06 thumb arthtiris spica is made from breathable AeroSpace™ material, which wicks away moisture, prevents overheating, and ensures comfort even during prolonged wear.
A system of adjustable hook-and-loop straps allows for individualized adjustment of stabilization and compression. The anatomical shape of the product ensures a precise fit to the natural contours of the hand and thumb, effectively reducing pressure on sensitive areas and bony prominences.
The AM-D-06 saddle joint stabilizing brace effectively maintains the thumb in its anatomical axis, limits painful movements, and reduces stress on joint and ligament structures. The product supports the healing and regeneration process in inflammatory conditions and after injuries.
The thumb stabilizer splint may be worn during the day, both during everyday activities and during work requiring repetitive hand movements.
The product is recommended both for conservative treatment and during recovery after injuries and surgical procedures involving the thumb and wrist.
Purpose of use:
- degenerative changes of the thumb carpometacarpal joint (rhizarthrosis),
- inflammatory and overload conditions of the thumb,
- thumb ligament injuries (e.g. skier’s thumb),
- instability of the thumb joint,
- overuse-related wrist pain,
- post-operative and post-traumatic periods (as recommended by a physician or physiotherapist),
- injury prevention during work and manual activities.
Sizes
| Size | Wrist circumference | How to measure |
| M | 13-17 cm |  |
| L | 17-22 cm |
Fits for both hands.
Total length of the product:
M: 23 cm
L: 25 cm
Gallery
Technology
MATERIALS
AeroSpace™
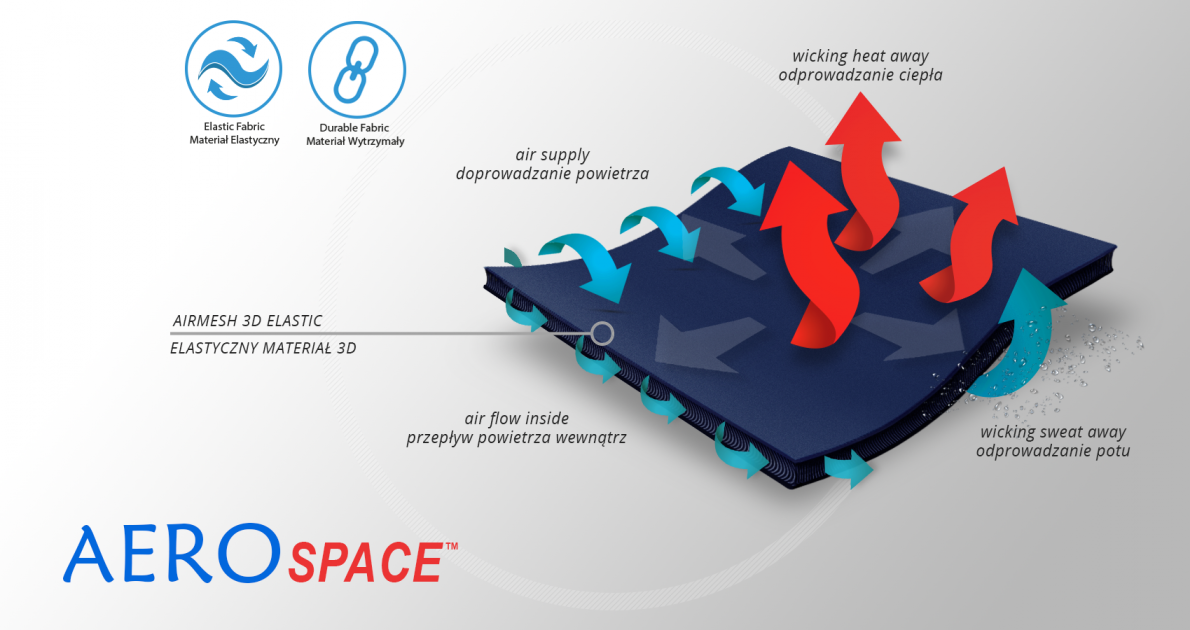
AeroSpace™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.