OKD-40

 Knee brace
Knee brace Breathable
Breathable Class I medical device
Class I medical device Double-sided
Double-sided Polycentric joint 2
Polycentric joint 2OKD-40 – PATELLA KNEE SUPPORT BRACE WITH DUAL-AXIS HINGES
Description
KNEE SPRAIN AND PATELLAR INSTABILITY
A knee sprain is an injury to the soft tissues of the joint, most commonly affecting the ligaments, joint capsule, or periarticular structures. It usually occurs as a result of a sudden twisting movement of the limb with the foot fixed on the ground and may lead to pain, swelling, and limited range of motion. Knee sprains are often accompanied by patellar instability, which involves an abnormal tracking of the patella. This condition may cause a sensation of the knee “giving way,” anterior knee pain, and recurrent dislocations.
Such conditions frequently require the use of a knee and patella-stabilising brace such as the OKD-40, which supports proper patellar tracking, reduces pain, and improves safety during movement.
Product’s description:
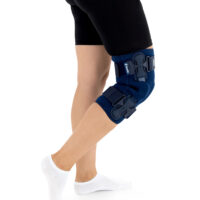
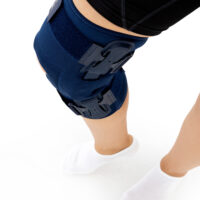
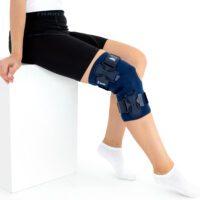
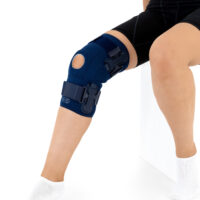
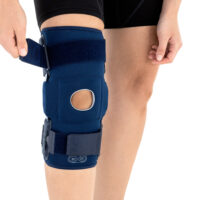
The OKD-40 professional knee brace provides stabilisation and controlled rehabilitation of the knee joint, with particular emphasis on patellofemoral stability.
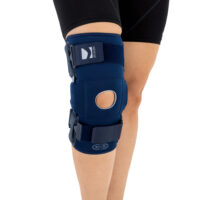
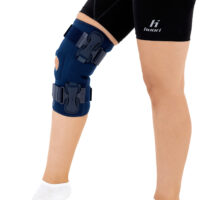
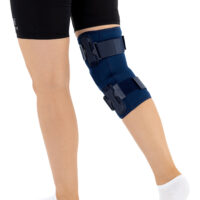
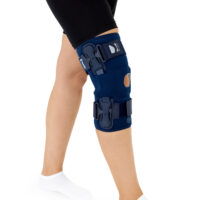
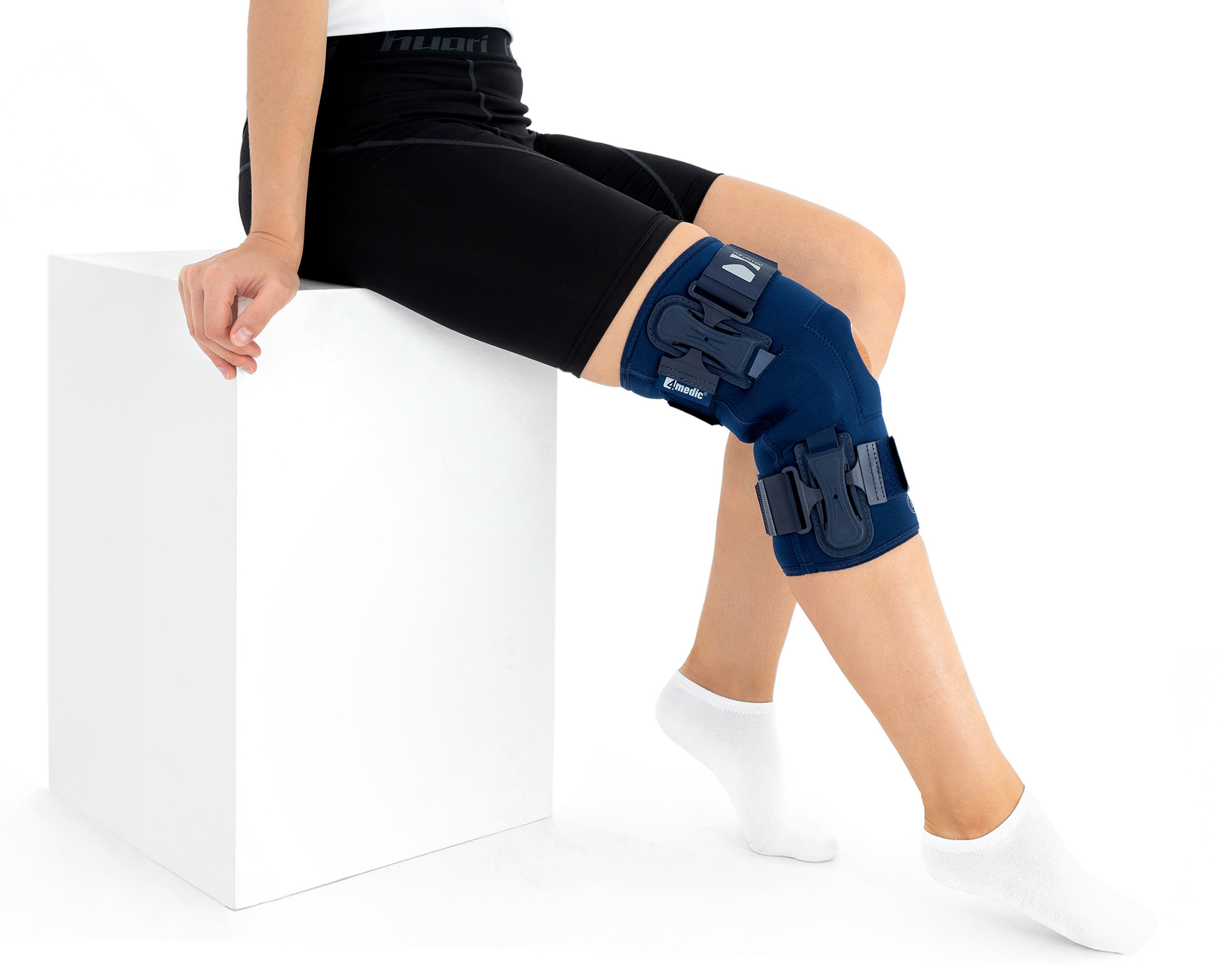
The knee brace features a closed design, fully encompassing the knee joint, thigh, and lower leg. This structure increases lateral and anterior–posterior stability, prevents the brace from shifting during movement, and ensures even compression.
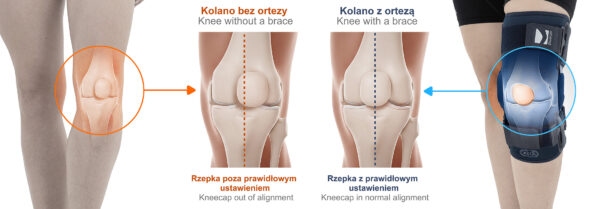
The OKD-40 hinged knee brace includes an anatomical patellar donut, which stabilizes the patella, maintains its correct tracking, and reduces patellofemoral overload. Thanks to this feature, the brace effectively reduces pain, prevents lateral patellar maltracking, and supports the treatment of chondromalacia and other conditions related to abnormal patellar alignment.
A detailed illustration of a human knee joint, highlighting areas of inflammation and pain. Suitable for medical publications, educational materials, or healthcare promotions.
OKD-40 professional knee brace is equipped with durable lateral splints with dual-axis hinges for ensuring anatomical axial alignment of the knee and protect against uncontrolled joint displacement. The applied dual-axis knee hinges replicate the natural biomechanics of the knee joint, where rolling and gliding movements occur simultaneously during flexion and extension. The presence of two axes allows for improved control of this complex motion.
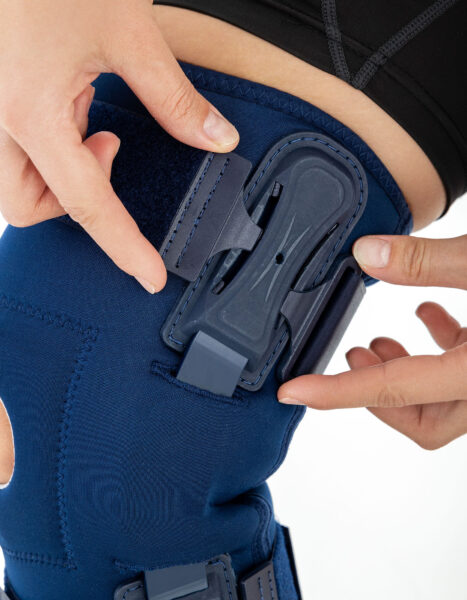
The hinges are integrated with the patented EASY FIT SOLUTION system, developed to further enhance the fit of orthopaedic braces to the anatomical contours of the body and the individual therapeutic needs of the user.
Manufactured from breathable, skin-friendly materials, the brace allows effective moisture management and prevents overheating, ensuring all-day comfort.
The OKD-40 patella knee support brace with dual-axis provides joint stabilisation while preserving natural knee biomechanics. It supports the treatment of knee sprains, patellar instability, and the rehabilitation process following injuries or orthopaedic procedures, increasing safety, comfort, and movement control during daily activities.
Purpose of use:
- patellar instability (kneecap lateralization)
- patellar chondromalacia
- knee osteoarthritis
- anterior knee pain
- patellar clicking
- patellofemoral joint inflammation
- ACL, PCL, LCL, and MCL injuries
- knee sprain
- meniscus injuries
- soft-tissue injuries
- postoperative stabilization
Sizes
| Size | Knee circumference | How to measure |
| S | 30 – 34 cm | 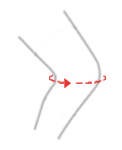 |
| M | 34 – 38 cm | |
| L | 38 – 42 cm | |
| XL | 42 – 46 cm | |
| 2XL | 46 – 50 cm |
Fits for both knees.
Total length of the product:
S – 2XL: 32 cm
Gallery
Technology
MATERIALS
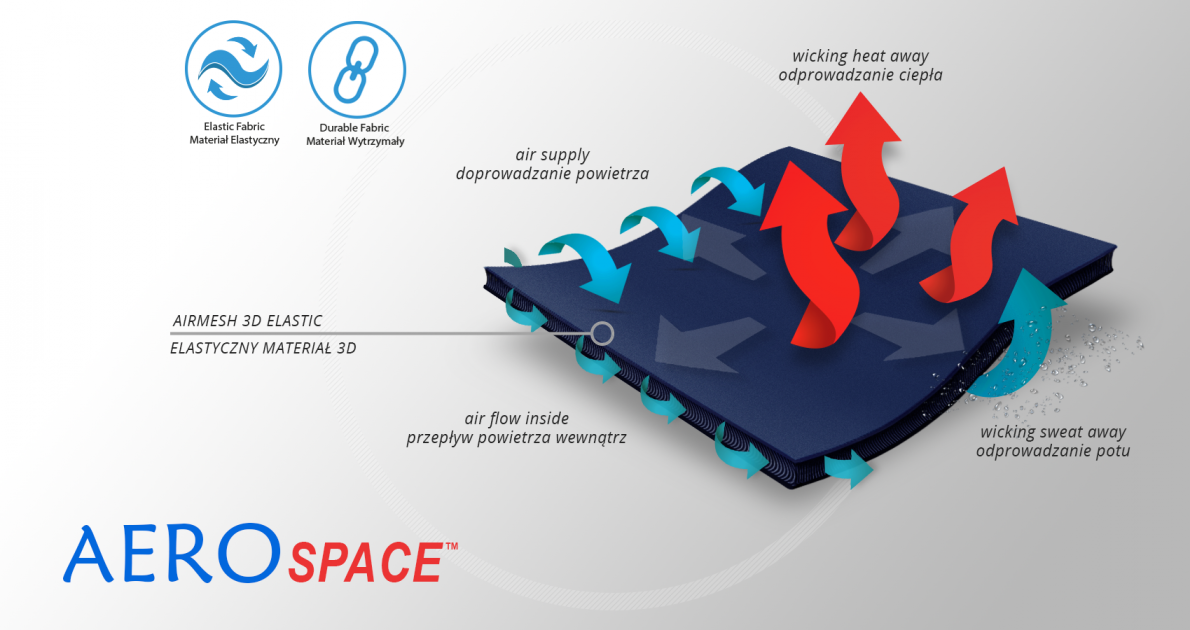
AeroSpace™
AeroSpace™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.
TECHNOLOGICAL SYSTEMS
EASY FIT Solution
 The patented EASY FIT SOLUTION system is designed to enhance the fit of products to body curves and therapeutic needs of users. The system is intended for injury stabilization and posture correction.
The patented EASY FIT SOLUTION system is designed to enhance the fit of products to body curves and therapeutic needs of users. The system is intended for injury stabilization and posture correction.
It consists of universal inserts, which allow for the attachment of clips with elastic elements of the stabilization system and provide angular movement, enabling precise adjustment of products to the user's body curves.
Additionally, the inserts allow for circumferential guidance of straps, providing circumferential stabilization of the selected area. The system is attached to soft components of products such as lower and upper limb orthoses, vests, or circumferential belts.
EASY FIT SOLUTION enables combining rigid components (splints, bars) with elastic straps, providing an individual approach when using standard rehabilitation equipment.
STIFFENINGS
Splints [2W]
Splints [2W] - polycentric, dual-axis hinges equipped with a rack-and-pinion mechanism, designed to ensure that flexion and extension closely replicate the anatomical movement of the knee joint. These hinges are primarily used in knee braces in cases where a high level of lateral joint stabilization is required. They are made of a high-quality aluminum alloy, making them lightweight, resistant to the effects of perspiration and salt, and sufficiently rigid to ensure proper performance of the device.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.