AM-OSN-U-15

 Wrist brace
Wrist brace Anatomic wrist splint
Anatomic wrist splint Breathable
Breathable Cast replacement
Cast replacement Class I medical device
Class I medical device Double-sided
Double-sided ER
ER Full range of adjustment
Full range of adjustment Zespół cieśni nadgarstka
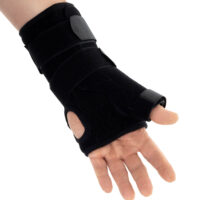
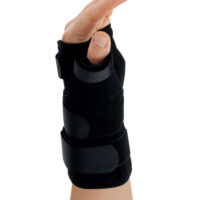
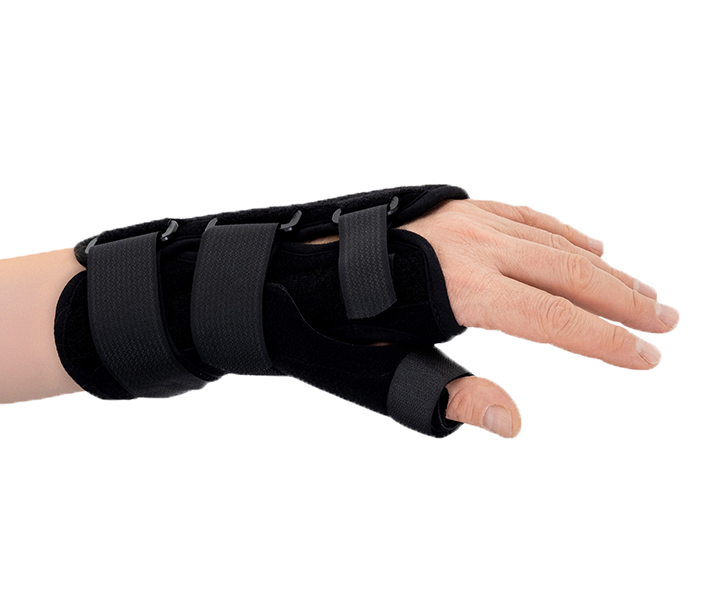
Zespół cieśni nadgarstkaAM-OSN-U-15 –WRIST BRACE WITH A DETACHABLE THUMB SPLINT
Description
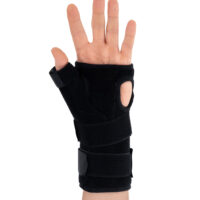
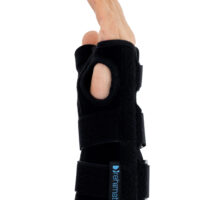
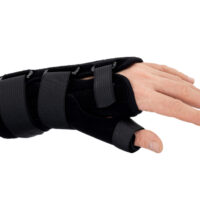
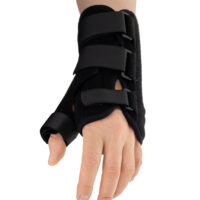
The wrist and forearm brace with a detachable thumb splint is a medical device intended for the stabilisation and unloading of the wrist joint and thumb joint.
The AM-OSN-U-15 wrist brace has been designed for users with injuries or disorders of the wrist joint and/or thumb, such as sprains, dislocations, inflammatory conditions, overload syndromes, or during the healing process. The product limits the range of motion of the wrist and thumb, helping to reduce pain, protect osteoligamentous structures, and support the treatment and rehabilitation process. It provides therapeutic and stabilising support while taking into account smaller sizes and increased comfort.
The wrist brace may be used prophylactically, conservatively, after injuries, as well as during the recovery period following cast immobilisation. The removable thumb splint allows for individual adjustment of the level of immobilisation according to the patient’s current therapeutic needs.
The AM-OSN-U-15 wrist brace features an innovative design, and the anatomically contoured palmar splint stabilises the radiocarpal joint in a neutral position, limiting unwanted movements and supporting regeneration.
Depending on therapeutic requirements, the lateral thumb stabiliser can be easily attached to the orthosis, providing professional immobilisation of the carpometacarpal (CMC) joint of the thumb.
The AM-OSN-U-15 wrist splint is made of skin-friendly, breathable materials that ensure continuous therapeutic compression of the joint. High-quality materials minimise the risk of skin irritation and improve wearing comfort, even during prolonged use.
The wrist brace offers a wide range of adjustment using hook-and-loop fasteners, allowing it to fit various wrist sizes and enabling quick and easy application and removal, which is particularly important for user’s needs.
Purpose of use:
- Wrist sprains and strains
- Ligament and tendon injuries
- Joint/tendon inflammation (e.g. caused by overuse)
- Rehabilitation after cast immobilisation
- Thumb stabilisation after injuries or in painful conditions
Sizes
| Size | Wrist circumference | How to measure |
| S | 11-13 cm |  |
| M | 13-17 cm | |
| L | 17-21 cm |
Fits for both wrists.
Total length of the product:
S-L: 21 cm
Gallery
Technology
MATERIALS
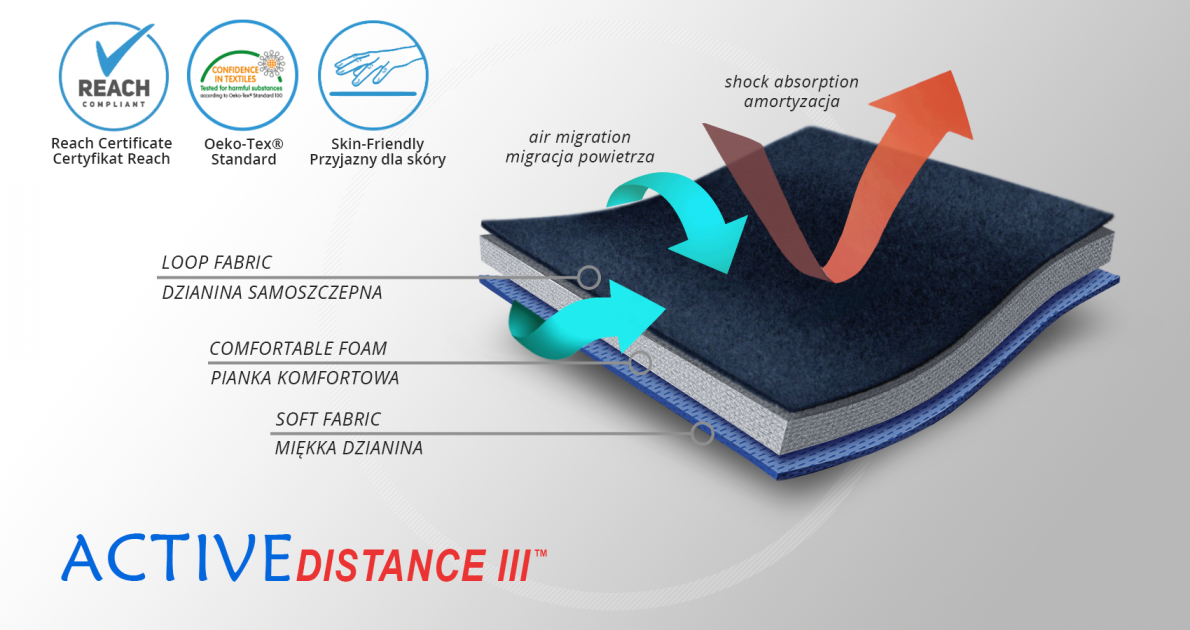
ActiveDistance III™
ActiveDistance III™ is an active 3-layered fabric made of non-elastic polyamide self-gripping layer, comfort foam and soft fabric. What is important, ActiveDistance III™ is non-elastic fabric so it stabilizes your body in a perfect way. Thanks to using the comfort foam, the device is really soft what influences on the comfort of using it. The last layer is responsible for maintaining your skin dry.
UniTape™
UniTape™ is a technological orthopedic lamination with high durability with double-sided self-gripping function. UniTape™ is very thin and easy to attach what allows to fit the braces precisely.
STIFFENINGS
Plastic stays
They come in various widths and thicknesses, are made of various types of plastics, such as polyamide, ABS or acrylic, and these features determine their stiffness. Thanks to their design, they are resistant to water, moisture and sweat. Products equipped with them can be washed without having to remove them from the orthosis. Our plastic stays work only in one direction, perfectly stabilize the laterally protected part of the body, adjusting to it at the same time and have a memory function, thanks to which they always return to their original shape. This function causes the stays in the orthosis to stabilize the swollen limb immediately after the injury and after the swelling has come off. The plastic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

Thumb splint
Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.