Leg support AS-U-02
THIGH BRACE WITH TRIAL COMPRESSIVE SYSTEM OF STRAPS
Description
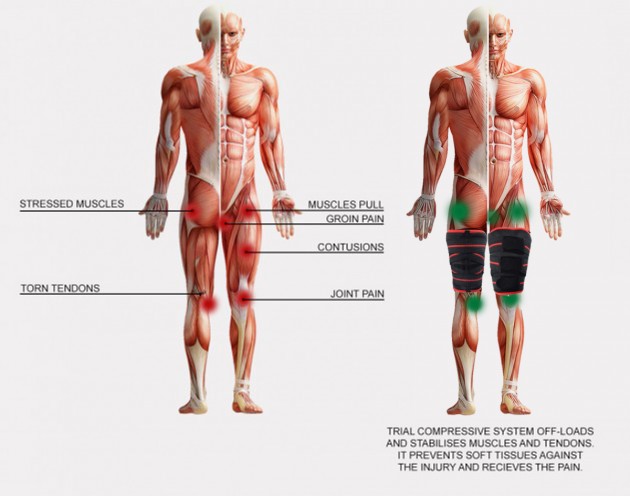
Hamstring strain
Sprinters, hurdlers, jumpers often suffer from hamstring injuries such as sprains, strains or ruptures. This condition occurs also in other sports with rapid speed (football, field hockey). Hamstring sprain is the most common injury in sprinters and has non-contact reasons.
Hamstring muscles are three posterior thigh muscles: semitendinosus, semimembranosus and biceps femoris. They connect two joints: hip and knee, so they predispose to injuries. The hamstring adduct and extend the hip and flex the knee.
Hamstring strain is the most common condition in Australian football player – 15% of all of the players in national league. Statistically, the injury occurs 6 times during one season and causes absence of athletes in 21 matches1. Similar situation is in British League, where hamstring injuries are 12% all of the conditions2. Ordinary player misses about 18 training weeks and can’t be part of 3 matches3.
Biceps femoris predisposes to injury, especially in musculotendinous area. Sprint bases on eccentric muscle contraction in loading response (LR) and then, after initial contact (IC), the muscle have concentric contraction. Hamstring strain occurs during eccentric work, when player doesn’t feel the ground.
There are a few symptoms of strain: pain, function disturbances, limited elasticity of muscles, hematoma and active trigger points.
After injury, compressive treatment is necessary, providing by our thigh brace AS-U-02.
Product’s description
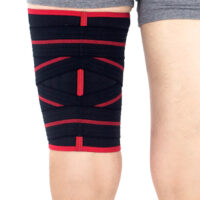
Our thigh brace supports and prevents against the injury both hamstring and quadriceps muscles.
This is anatomic-shaped thigh sleeve with trial compressive system of straps, equipped with Velcro.
Unique Y-shaped straps provide additional adjustable compression and stabilisation.
Our thigh sleeve AS-U-02 is made of AeroSpace™ and AirRubber II™
Anatomic-shape of the sleeve allows to use it for right and left leg.
Designed for a perfect compression according to your needs.
Purpose of use
– quadriceps / hamstring pull
– bruise of thigh muscles
– thigh muscles strain
– trigger point in thigh area
– avulsion of muscle insertions
– poor blood flow
__________________
1 Orchard J, Steward H., Epidemiology of injuries in the Australian Football League, seasons 1997-200. Br J Sports Med. 2002; 36: 39-45.
2 Hawkins RD, Fuller CW. A prospective epidemiological study of injuries on four English professional football clubs. Br J Sports Med. 1999; 33: 196-203.
3 Woods C, Hawkins RD, Maltby S, et. Al. The football association medical research programme: an audit of injuries in professional football – analysis of hamstring injuries. Br J Sports Med. 2004; 38: 36-41.
Sizes
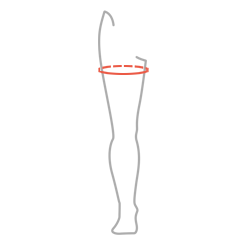
| Size | Thigh circumference 5 cm below the groin | How to measure |
| S | 48-52 cm (18,9″-20,5″) |
 |
| M | 52-56 cm (20,5″-22″) |
|
| L | 56-60 cm (22″-23,6″) |
|
| XL | 60-64 cm (23,6″-25,2″) |
Total length of the product: 26 cm (10.2″)
Gallery
Technology
MATERIALS
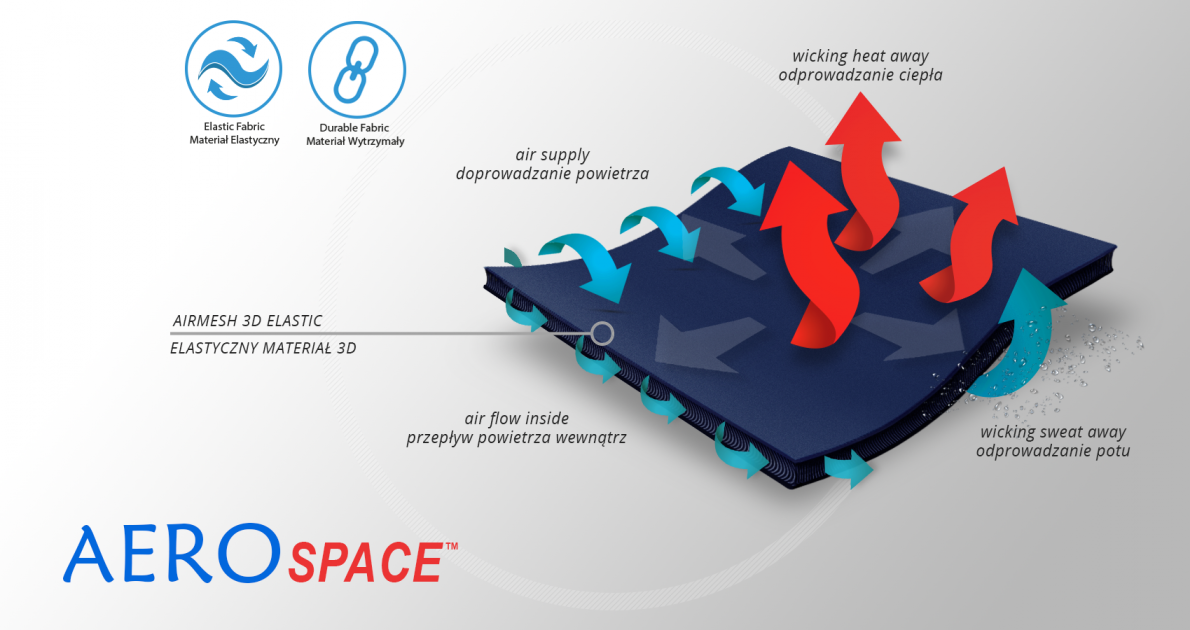
AeroSpace™
AeroSpace™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.
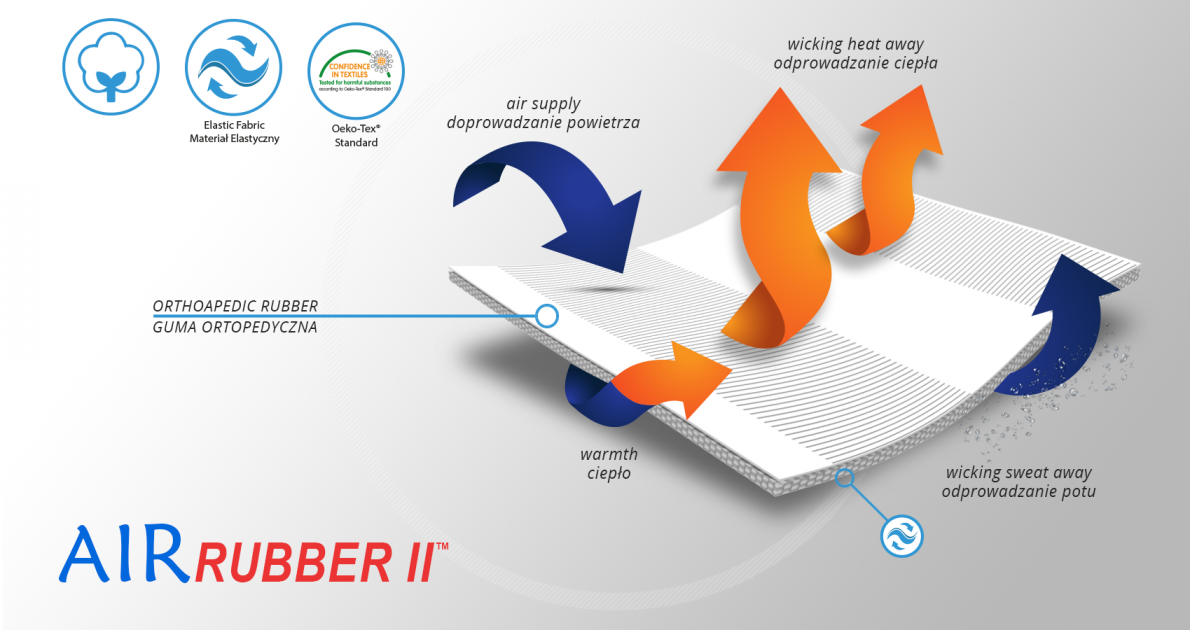
AirRubber II™
AirRubber II™ is breathable, perforated and has unidirectional elasticity. It can be stretched, increasing the length, not width, what improves compression. Between the braids air flows freely and skin can breathe. In addition in high compression, these rubber braids provide a gentle massage for skin. Our orthopedic rubber is very friendly for skin.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.