OSS-OS-06

 Foot brace
Foot brace Class I medical device
Class I medical device Compensation
Compensation Foot drop
Foot drop Innovative
Innovative Invisible
Invisible Neurology
Neurology Recommended by specialists
Recommended by specialists Waterproof
WaterproofOSS-OS-06 – FOOT DROP BRACE FOR WALKING
Description
FOOT DROP
Foot drop is a condition in which the patient has difficulty lifting the front part of the foot during walking. It most commonly results from weakness or damage to the peroneal nerve, neurological disorders, injuries, stroke, or dysfunction of the muscles responsible for dorsiflexion. Individuals with this condition often drag their toes along the ground, increasing the risk of tripping and falling, while gait becomes unstable and tiring.
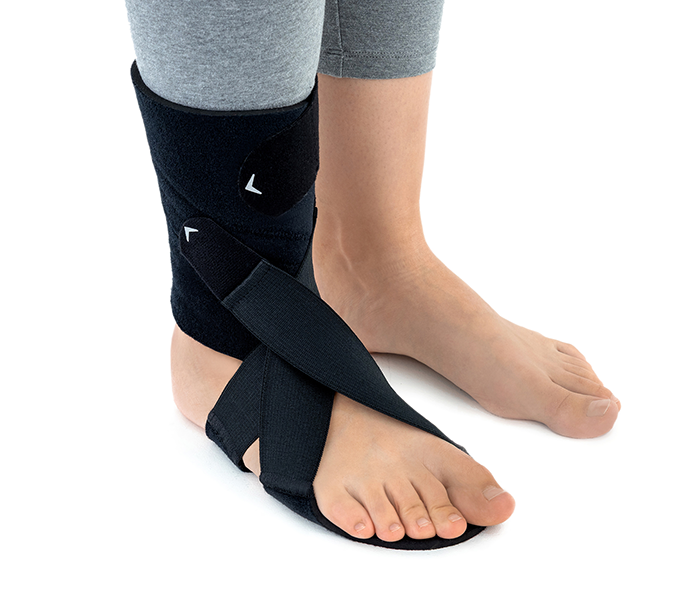
In such cases, the OSS-OS-06 foot drop brace for walking lifts the front part of the foot and supports a proper gait pattern, improving safety and overall walking comfort.
Product’s description:
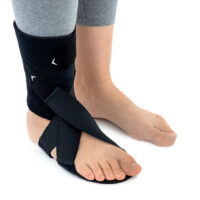
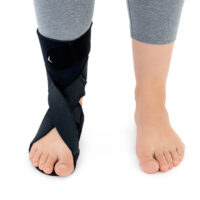
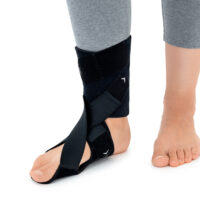
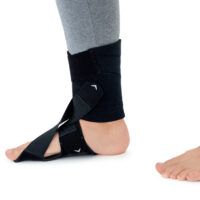
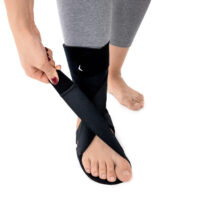
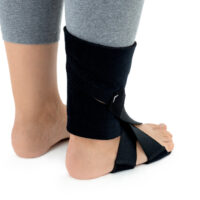
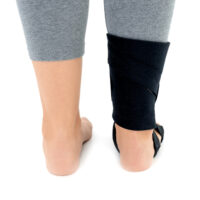
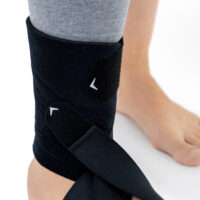
The OSS-OS-06 foot drop splint is designed to effectively elevate the forefoot in patients with foot drop. The product also provides functional support following injuries, in muscle weakness, or neurological disorders.
This wrap-around foot brace with the X-shaped Velcro closure allows full adjustment to the anatomy of the lower limb. The fastening system is equipped with a silicone lining that prevents the straps from shifting during use.
Elastic compression straps ensure smooth adjustment of the dorsiflexion angle within a range of 45°–135°. The brace maintains the ankle in a neutral 90° position while gradually lifting the forefoot, making walking easier and providing continuous stretching of the plantar tissues and the Achilles tendon. Lifting the toes prevents the foot from dragging across the ground, which is crucial in foot drop therapy.
The plantar section of the ankle brace is equipped with a plastic reinforcement that effectively prevents foot drop, while its dynamic properties support an efficient push-off phase during gait.
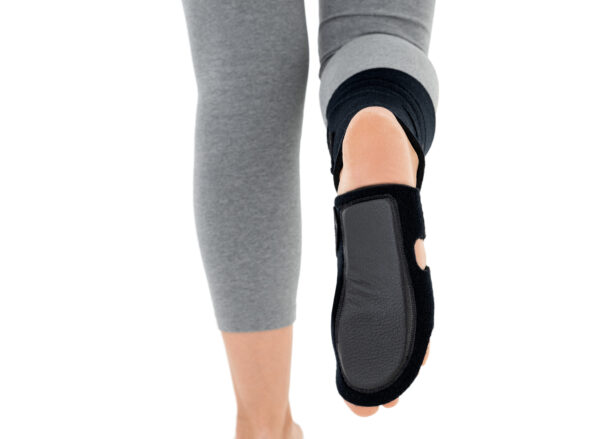
The OSS-OS-06 foot drop brace for walking is made of breathable, lightweight UniPren material, ensuring comfort during daily use. The brace can be worn with or without footwear.
The brace reduces foot pain and stiffness, improves gait safety in cases of foot drop, supports the physiological pattern of toe lift, enhances mobility, and increases overall walking comfort. It is an excellent complement to gait therapy for neurological and orthopaedic patients.
The OSS-OS-06 foot drop brace for walking has universal application and may be used in cases of foot drop, Achilles tendon tightness, plantar fascia inflammation, overload conditions, and nerve injuries.
Purpose of use:
- Foot drop
- Weakness or paralysis of the peroneal nerve
- Neurological conditions (stroke, neuropathies, MS, ALS)
- Plantar fasciitis
- Achilles tendon tension or inflammation
- After injuries or surgeries of the foot or ankle
- Muscle weakness and gait disturbances
- Support in gait rehabilitation therapy
Sizes
| Size | Foot length | Ankle circumference | How to measure |
| M | 22 – 26 cm | min 20 cm – max 33 cm | 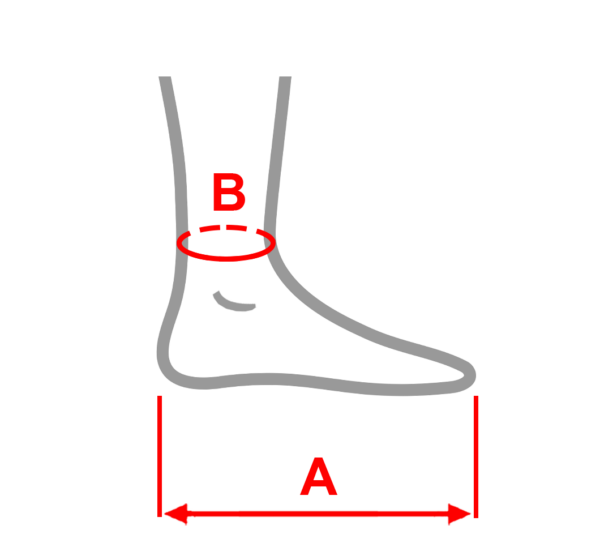 |
| L | 26 – 30 cm |
Right and left foot specific.
Total height of the product:
M-L: 22 cm
Gallery
Technology
MATERIALS
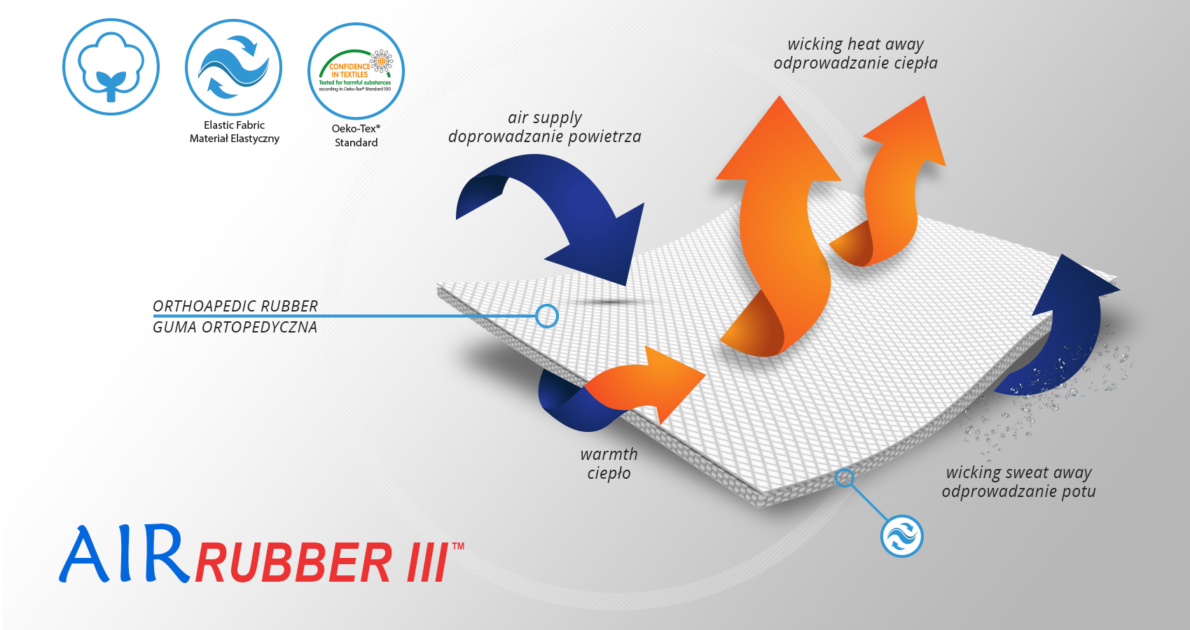
AirRubber III™
AirRubber III™ has unidirectional elasticity. It can be stretched, increasing the length, not width, what improves compression. Between the braids air flows freely and skin can breathe. In addition in high compression, these rubber braids provide a gentle massage for skin. Our orthopedic rubber is very friendly for skin.
UniPren™
UniPren™ is a universal 3-layer material consisting of an external elastic polyamide cover knit with a self-adhesive function, an internal neoprene foam core and an elastic jersey cover knit. This material is characterized by softness and very high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of UniPren™ are the strongest and most effective stabilizing orthoses available on the market. Self-adhesive function, the raw material makes it much easier to use.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.