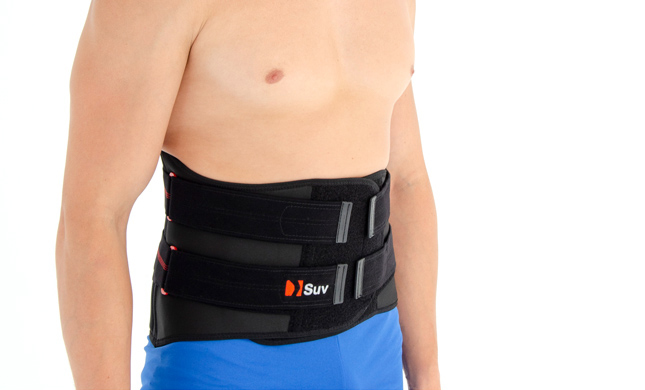
LOWER BACK BRACE AM-SO-04

 LSO
LSO Anatomic stays
Anatomic stays Breathable
Breathable Class I medical device
Class I medical device Compression
Compression Innovative
Innovative Neurology
Neurology Orthopedics
OrthopedicsSUV
MALE LUMBAR BACK BRACE WITH SUPPORT STRAPS
Description
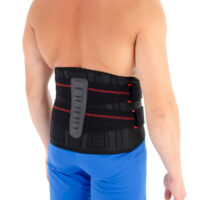
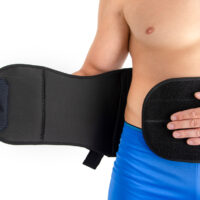
AM-SO-04 lumbar back support with fasteners for men – SUV was developed according to anatomical shape and provides adequate level of stabilization on lumbar area.
The Back part of support is made of innovative Active3D™.
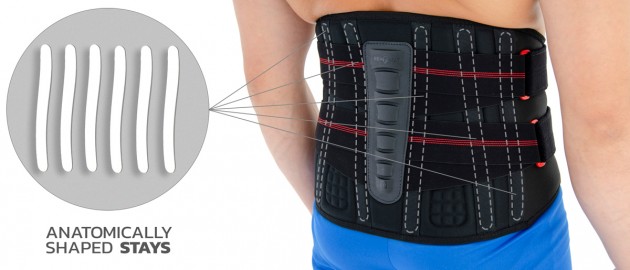
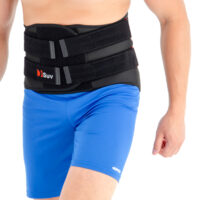
The orthosis is provided with anatomically shaped metal stays (sweat-resistant coating), which quantity depends on the size; 4 or 6 metal stays in the Back. The orthosis incorporates semi-rigid Back Plate which function is, to provide reinforcement of lumbar stabilization.
The lateral parts are made of AeroSpace™.
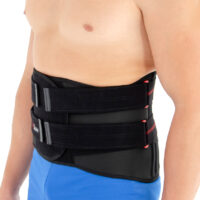
An asymmetric closure allows the perfect fitting on the patient body. The panel of closure is made of velcro laminate with comfortable semi-rigid foam located inside. The system GRIP & OPEN makes the orthosis easy to assemble and disassemble.
Easy to apply due to the closure system with hand grip pockets, especially developed for patients with hands disability.
The system of double tensioners made of AirRubber III™ stabilizes and regulates compression in the up and down part of orthosis, allowing improvement in adjustment and fitting to the patient´s body.
The back fasteners connected to the Lumbar Plate provide compression on lumbar area.
Optionally, the orthosis AM-SO-04 could be provided with:
– Suspenders – useful during the physical training or work.
Properties
– SKIN FRIENDLY PRODUCT – lumber support has been made of special orthopaedic rubber, containing mainly cotton, which is neutral to patient’s skin
– EFFECTIVENSS OF STABILIZATION – the stabilization is exceptionally good thanks to the orthopaedic nibs, tightening rubber belts crossed at the back and anatomically shaped loin pillow.
– SAFETY – the front tightening rubber belts provide additional protection and the chances of the device to undo accidentally are eliminated.
– COMFORT OF USAGE – the limber support is very light and therefore comfortable to wear.
Indications
– Pre- and Post-Surgical Stabilization
– Degenerative Spinal pathologies on Lumbar area
– Disc Hernia
– Lumbar Muscles and Ligamentous Strains and Sprains
– Postural Support
– Chronic Low Back Pain
– Rehabilitation and Prevention
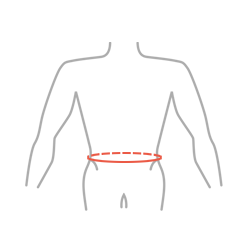
Sizes
| Size | Waist circumference | How to measure |
| S | 65-75 cm (25,6″-29,5″) |
 |
| M | 75-85 cm (29,5″-33,5″) |
|
| L | 85-97 cm (33,5″-38,2″) |
|
| XL | 97-110 cm (38,2″-43,3″) |
|
| XXL | 110-125 cm (43,3″-49,2″) |
|
| XXXL | 125-145 cm (49,2″-57,1″) |
Total height of the product:
front: 19 cm (7,5″)
tył: 28 cm (11″)
Gallery
Technology
MATERIALS
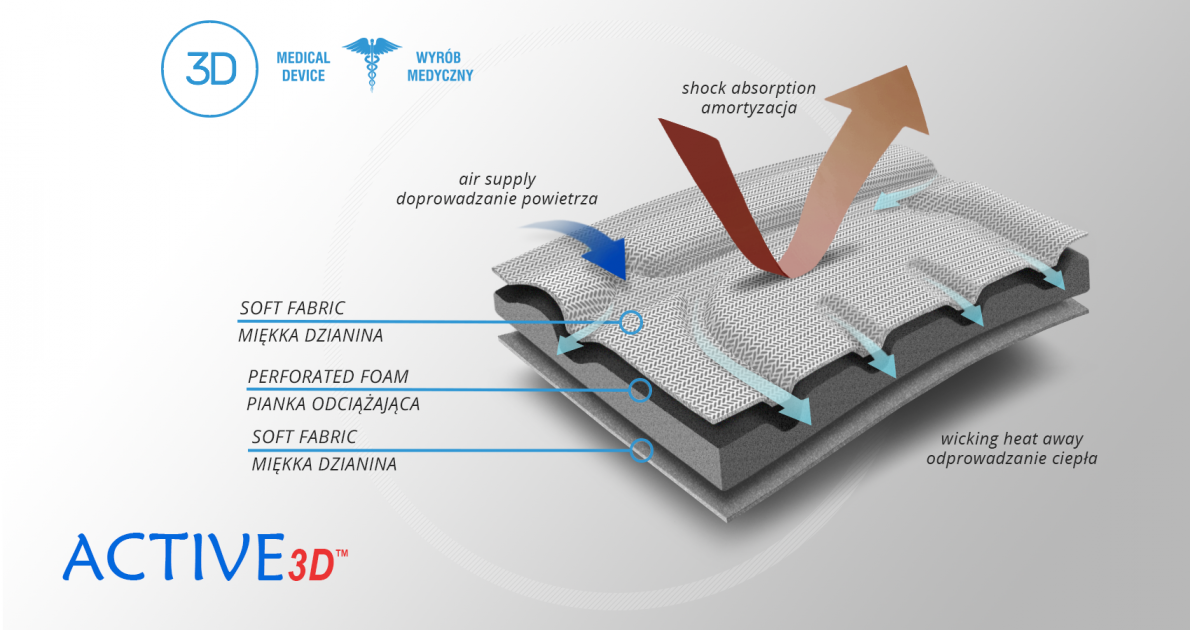
Active3D™
Active3D™ is thermoformed fabric made of special foamed, cell-closed designed material. It reduces the pressure on the body or any abrasions made by orthopaedic stays and aluminum splints. It is fully waterproof fabric and does not absorb sweat. It’s easy to clean. Due to its features, the fabric is an excellent product for making medical orthopaedic braces and orthoses. ACTIVE 3D™ has various external self-gripping layers. Our material has special, thermoformed properties and may be shaped according to the functional goals of the final braces.
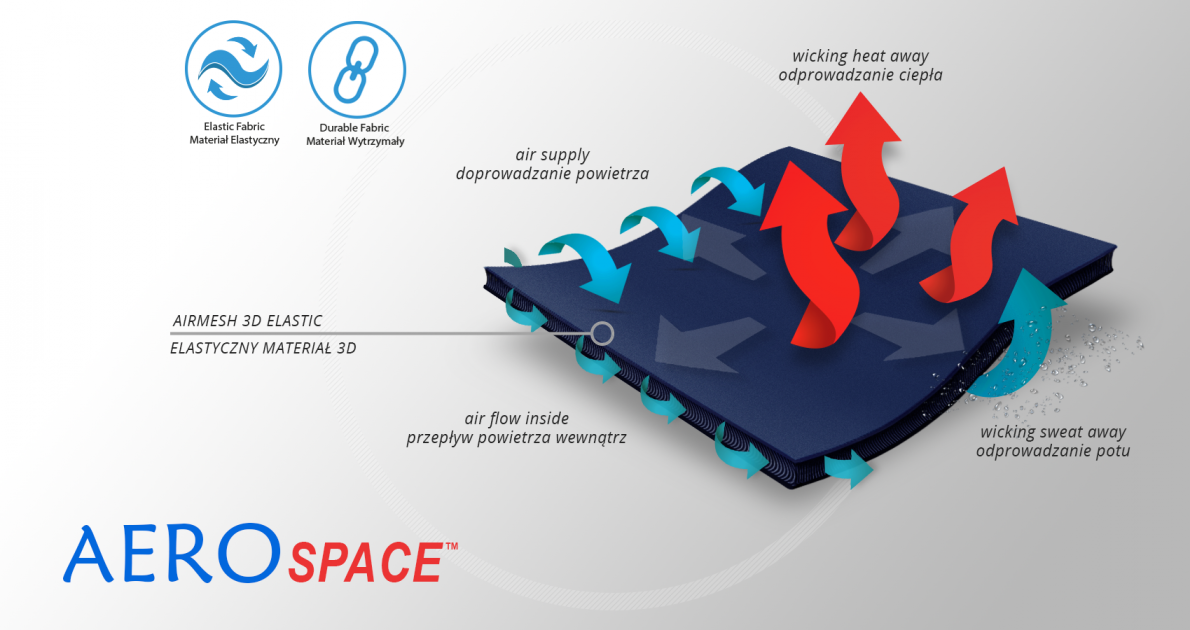
AeroSpace™
AeroSpace™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.
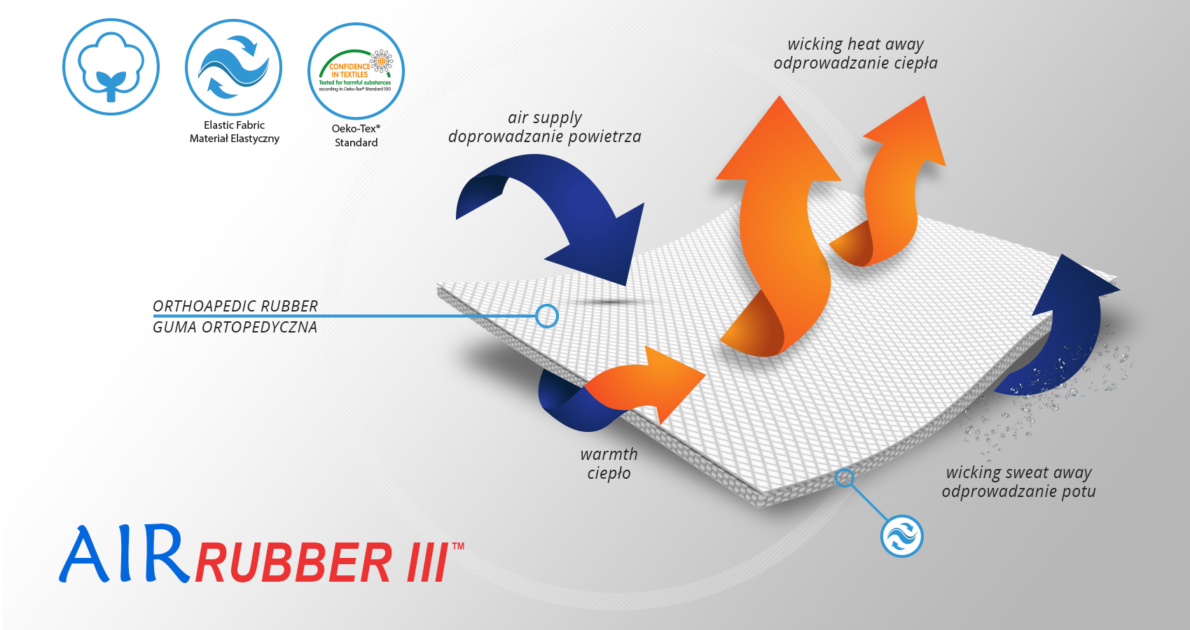
AirRubber III™
AirRubber III™ has unidirectional elasticity. It can be stretched, increasing the length, not width, what improves compression. Between the braids air flows freely and skin can breathe. In addition in high compression, these rubber braids provide a gentle massage for skin. Our orthopedic rubber is very friendly for skin.
STIFFENINGS
Lumbar supports
Low and high lumbar supports are made of polyethylene injection technology. The supports are embossed in the shape of the vertebrae of the spine, thanks to which it does not press on the spine, but relieves the back muscles. The product is equipped with the manufacturer's logo and a path for the seam. They stiffen the lumbar or thoracolumbar spine, relieve the muscle and stabilize the compression bands of the braces.

Orthopedic profiled stays
Orthopedic stays are made of special spring steel, covered with a plastic coating and have rounded and protected ends. They can come in various widths and thicknesses as well as different hardness. They can be pre-shaped or flat. They work in one direction and adapt to the shape of the body while stabilizing the laterally protected part of the body. They are perfectly protected against corrosion, so they are resistant to water, moisture and sweat. Products equipped with them can be washed without removing them from the orthosis. They adjust to the body. The orthopedic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

Orthopedic stays from ABS
Orthopedic stays available in various width, thickness and shapes are cut from ABS boards. Their role is not to stiffen the orthosis in order to support a joint or other part of the body, but to prevent wrinkling and uncontrolled movement of the fabrics of the orthosis./p>

Plastic stays
They come in various widths and thicknesses, are made of various types of plastics, such as polyamide, ABS or acrylic, and these features determine their stiffness. Thanks to their design, they are resistant to water, moisture and sweat. Products equipped with them can be washed without having to remove them from the orthosis. Our plastic stays work only in one direction, perfectly stabilize the laterally protected part of the body, adjusting to it at the same time and have a memory function, thanks to which they always return to their original shape. This function causes the stays in the orthosis to stabilize the swollen limb immediately after the injury and after the swelling has come off. The plastic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

PADDINGS
3D supports
3D relief supports are independent technical solutions to relieve the rigid elements of a given orthosis. These elements are made of supporting foams or EVA foam. These foams are connected with various types of skin-friendly materials and materials with an adhesive function. These pads have the appropriate shape and color adapted to the type of orthosis. They relieve both metal elements of orthoses, such as splints, stays, underwires and orthopedic drop locks, as well as other elements that should not come into direct contact with the patient's skin. These pads have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, guaranteeing the proper therapeutic effect.
Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.