Torso support OT-04


 TLSO+
TLSO+ Breathable
Breathable Class I medical device
Class I medical device Neurology
Neurology Orthopedics
OrthopedicsThoraco Lumbar Sacral Brace
Description
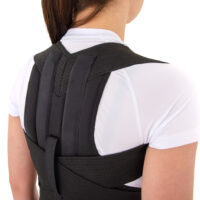
High thoraco lumbar sacral brace is very comfortable and functional. It is made of airy materials what allows sweat to evaporate from the skin. Anatomic shape provides good device fitting and better TLSO stabilization. Thanks to under armpit straps allow to achieve full anterior-posterior stabilization.
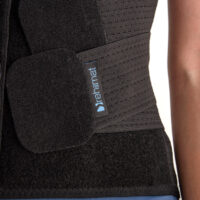
Lateral parts of the brace are made of perforated orthopaedic foam, which has only one-direction elasticity. The little wholes allow skin to breath.
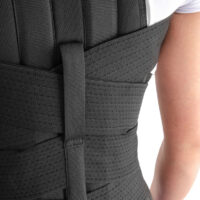
The rear part of the OT-04, depends on the size, is equipped with 2 or 4 orthopaedic splints and high aluminium spine chassis. The orthopaedic splints are anatomically shaped and hold sweat-resistant coating.
Two sets of double straps provide better thoracolumbar spine stabilization.
Indications
OT-04 should be applied in case of:
- severe L-S painful syndrome
- multilevel Degenerative Disc Disease
- multilevel discopathy
- osteoporosis
- spine instability
- lumbar spondylolisthesis
- Spinal Compression Fractures
- sciatica
- after surgeries (orthopaedics, neurology)
Sizes
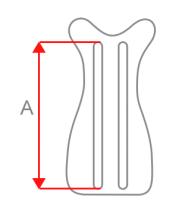
| Size | Waist circumference | Height (A) | How to measure |
|---|---|---|---|
| S | 65-75 cm (22,6″-29,5″) |
43 cm (16,9″) |
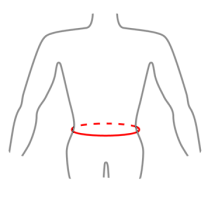  |
| M | 75-85 cm (29,5″-33,5″) |
46 cm (18,1″) |
|
| L | 85-97 cm (33,5″-38,2″) |
49 cm (19,3″) |
|
| XL | 97-110 cm (38,2″-43,3″) |
52 cm (20,5″) |
|
| XXL | 110-125 cm (43,3″-49,2″) |
55 cm (21,7″) |
|
| XXXL | 125-145 cm (49,2″-57,1″) |
| Size | Total height of the product | |
|---|---|---|
| rear | front | |
| S | 48 cm | 20 cm |
| M | 51 cm | |
| L | 54 cm | |
| XL | 57 cm | |
| XXL | 59 cm | |
| XXXL | ||
Gallery
Technology
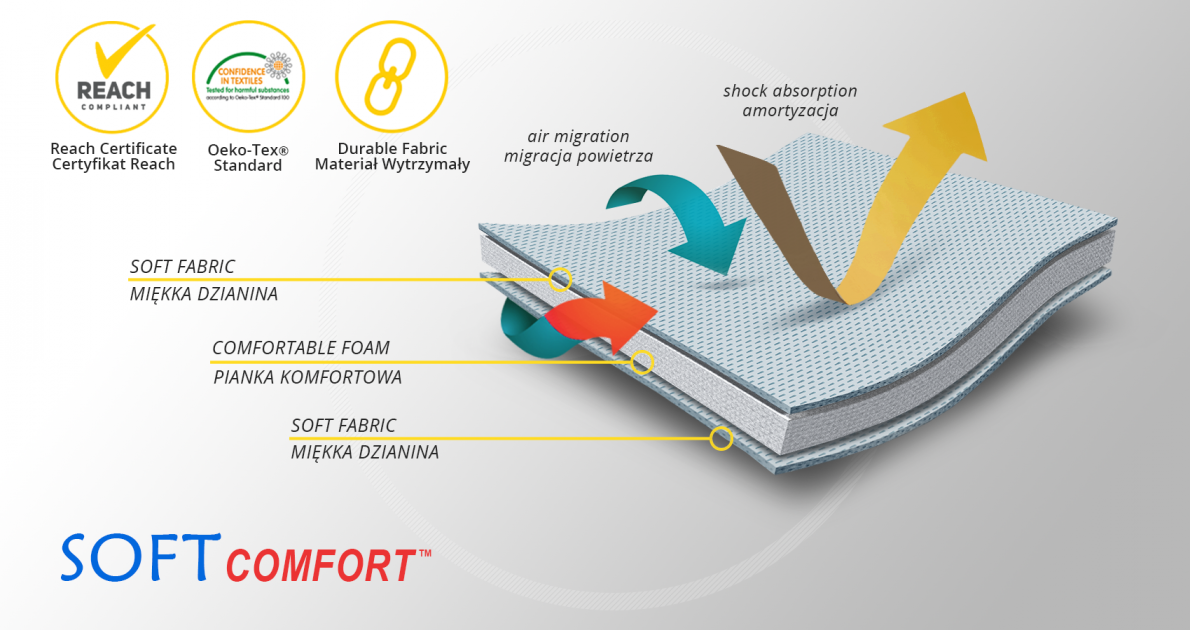
MATERIALS
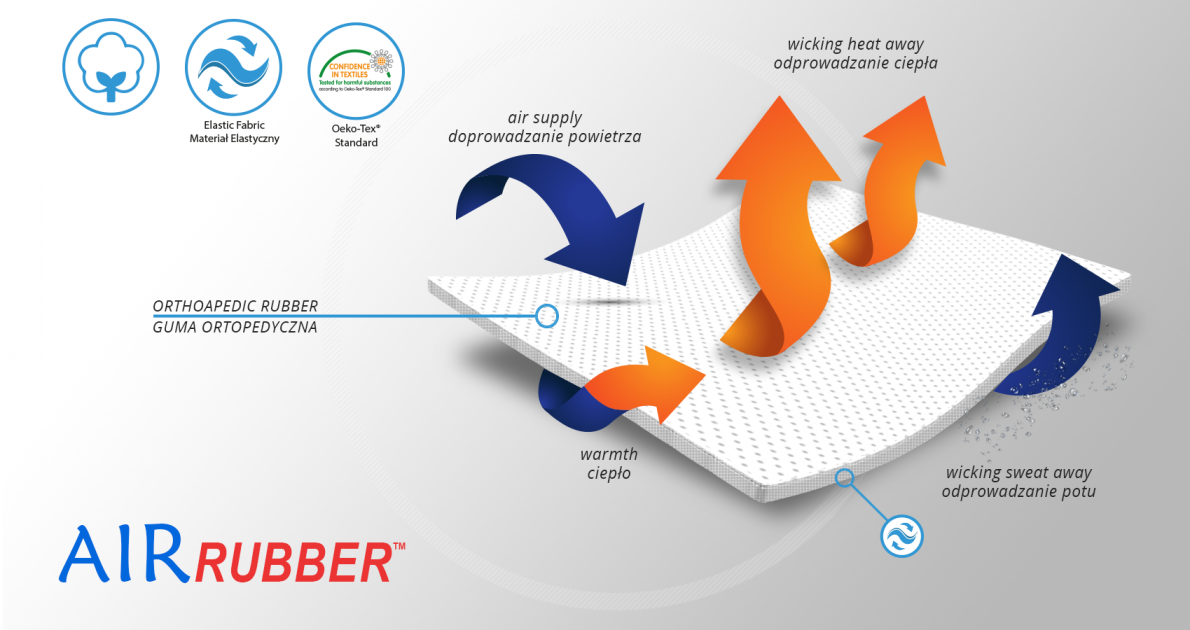
AirRubber™
AirRubber™ has unidirectional elasticity and it’s breathable. This lamination is perforated. Between the braids air flows freely and skin can breathe. In addition in high compression, these rubber braids provide a gentle massage for skin. Our orthopedic rubber is very friendly for skin.
SoftComfort™
SoftComfort™ - a material specially designed to relieve very delicate places on the patient's body. The cover knit used in the production is made in such a way that it can come into contact with very sensitive skin. This raw material has the Oeko-Tex Standard 100 certificate and complies with the European REACH standard. The structure of the material consists of a very soft comfort foam laminated with a delicate cover knit. The fabric has a perforation that facilitates the flow of air through the material and allows the skin to breathe. The SoftComfort™ material is perfect for the production of linings relieving the rigid and inflexible elements of orthopedic orthoses.
STIFFENINGS
Orthopedic profiled stays
Orthopedic stays are made of special spring steel, covered with a plastic coating and have rounded and protected ends. They can come in various widths and thicknesses as well as different hardness. They can be pre-shaped or flat. They work in one direction and adapt to the shape of the body while stabilizing the laterally protected part of the body. They are perfectly protected against corrosion, so they are resistant to water, moisture and sweat. Products equipped with them can be washed without removing them from the orthosis. They adjust to the body. The orthopedic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

Plastic stays
They come in various widths and thicknesses, are made of various types of plastics, such as polyamide, ABS or acrylic, and these features determine their stiffness. Thanks to their design, they are resistant to water, moisture and sweat. Products equipped with them can be washed without having to remove them from the orthosis. Our plastic stays work only in one direction, perfectly stabilize the laterally protected part of the body, adjusting to it at the same time and have a memory function, thanks to which they always return to their original shape. This function causes the stays in the orthosis to stabilize the swollen limb immediately after the injury and after the swelling has come off. The plastic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

Profiled aluminum stays
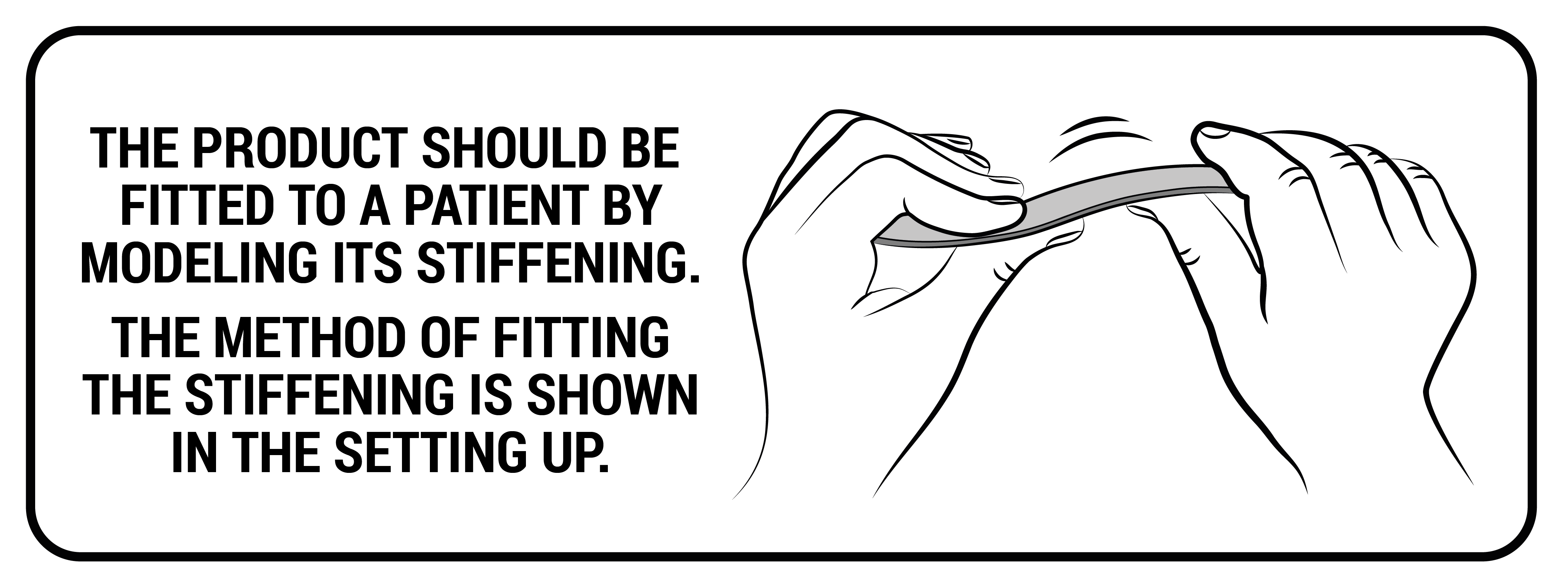
These are splints and orthopedic stays of various thickness and width, which are made of various types of aluminum alloys. All these splints and stays, before mounting to a given orthosis, have been pre-profiled, which allows for fitting the product to the body of a specific patient. However, for the correct operation of the device, they should be precisely bent to the patient's body by an orthopedist, physiotherapist or orthopedist technician. Only this action guarantees the proper protection and support of the patient's body.

Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.