Upper-extremity support OKG-10


 Hand brace
Hand brace Breathable
Breathable Class I medical device
Class I medical device Exercise ball
Exercise ball Innovative
Innovative Neurology
Neurology Skin-friendly
Skin-friendly Universal size
Universal sizeOKG-10 – ANTI-SPASTICITY HAND SPLINT WITH SEMI-BALL
Description
HAND SPASTICITY
Hand spasticity is a muscle tone disorder in which the muscles of the hand and fingers remain in a state of excessive, involuntary contraction. It most often results from damage to the central nervous system, e.g. after a stroke, injury, multiple sclerosis, or cerebral palsy. It leads to limited mobility, stiffness, pain, and difficulties in daily activities such as grasping or writing.
Prevention of spastic hand includes both active and passive therapy. Alongside rehabilitation and stretching exercises, it is recommended to use special wrist orthoses that maintain the hand in a neutral position, such as the OKG-10 type.
Product’s description:
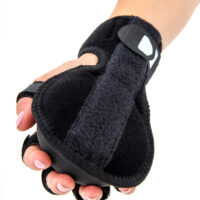
The OKG-10 anti-spasticity wrist brace with semi-ball was designed for individuals struggling with hand spasticity – due to stroke, hemiplegia, multiple sclerosis, dystonia, or contractures (e.g. Dupuytren’s contracture). It allows for comfortable and safe maintenance of the hand in a neutral position, reduces involuntary spasms, prevents deformities, and facilitates rehabilitation.
The brace has a lightweight, innovative design. Made from breathable AeroSpace II™ material, it prevents skin irritation and ensures continuous ventilation for all-day comfort.
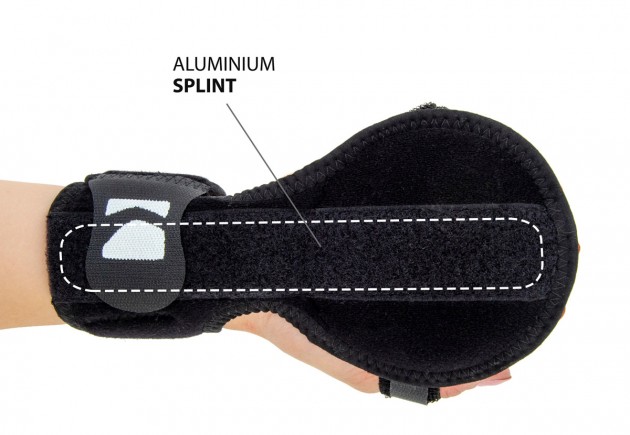
On the palmar side, an aluminium splint is integrated, effectively preventing palmar flexion and rotation of the hand.
Additionally, the OKG-10 hand splint features a built-in palm semi-ball, which applies pressure to the central part of the hand, relaxing the finger flexors and positioning the entire hand in a neutral, therapeutic posture.
Each finger has an independent Velcro strap, securing it individually and preventing the hand from “curling” into a fist.
Maintaining the hand in a functional gripping position prevents spasticity and supports performance of daily activities.
Regular use of the hand splint brace helps to keep the hand in a neutral position. It limits involuntary movements such as spasms and tremors. The semi-open hand position prevents contractures. Using the brace systematically during rest and at night helps to maintain joint mobility in the hand and fingers. Combined with rehabilitation and exercises, it supports functional recovery and reduces pain.
Indications:
- Finger flexion contracture
- Cerebral palsy
- Finger dysfunction
- Hemiplegia
- Finger stiffness
- Rheumatoid arthritis (RA)
- Psoriatic arthritis
- Muscular atrophy
- Stroke
- Haemorrhage
Sizes
| Size | Wrist circumference | How to measure |
| Universal | min 11 – max 24 cm min 4,3″ – max 9,4″ |
 |
Right and left hand specific.
Total length of the product: 21 cm (8.3″)
Gallery
Technology
MATERIALS
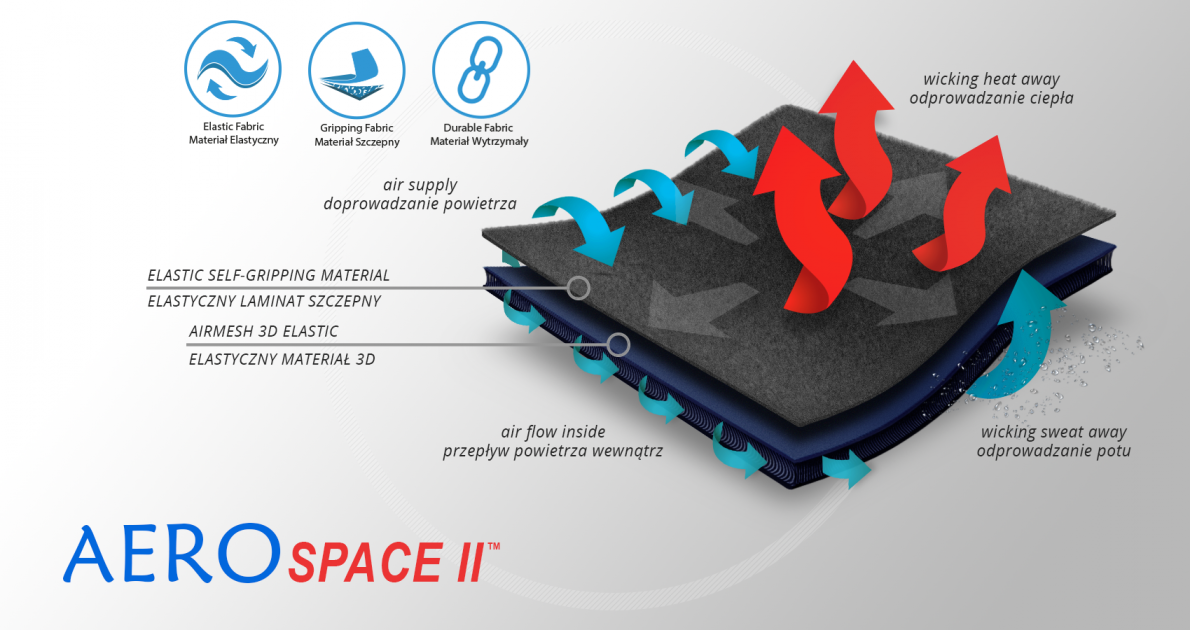
AeroSpace II™
AeroSpace II™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. AeroSpace II™ has an external self-gripping layer what makes it easy to adjust each product to individual patient’s needs. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.

STIFFENINGS
Flat aluminum stays
Aluminum stays are made of a special aluminum alloy that guarantees proper stiffness with minimal weight. They have rounded ends to prevent the damage of the product and come in various widths and thicknesses. The level of stabilization of the orthopedic device is defined by proper selection of the width and thickness of the aluminum stays. The stays can be pre-profiled or flat. They do not adapt to the shape of the patient's body, an individual adjustment of the orthosis is required by proper bending of the aluminum stays in the product. Thanks to this function, it is possible to correct the position of the patient's body or the secured joint.

PADDINGS
Molds made by injection method
PUR foam owes its popularity primarily to its physical and chemical properties, which are characteristic of polyurethane. The foam , depending on the proportion of the mixture, can be more rigid or flexible. Based on the physicochemical properties - density, and thus the stiffness or flexibility of the finished product - PUR can be used in an almost unlimited way.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.