Upper-extremity support OKG-22
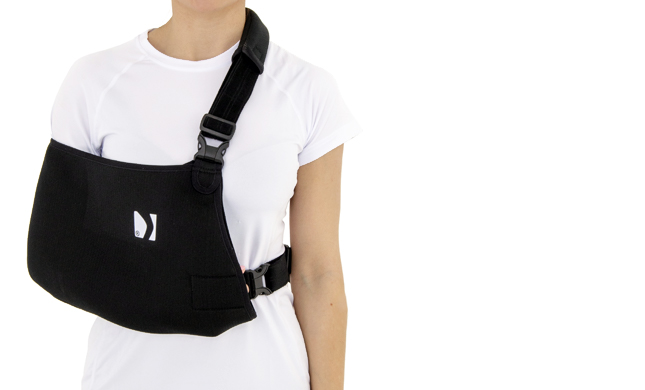

 Shoulder brace
Shoulder brace Upper limb brace
Upper limb brace Cast replacement
Cast replacement Class I medical device
Class I medical device Double-sided
Double-sided ER
ER Skin-friendly
Skin-friendlyOKG-22 THUMB-LOOP LEFT OR RIGHT SHOULDER SLING
Description
SHOULDER INJURIES
Shoulder injuries range from mild injuries i.e. sprains to more serious ones like fractures or dislocations. There are also frequent tendonitis or bursitis due to overuse of the joint. They usually occur as a result of overload, sports injuries or falls. The most common symptoms of damage to the shoulder joint include pain, stiffness, limited mobility and, in some cases, popping noises in the joint. Regardless of the cause, one of the primary forms of treatment for a shoulder injury is its immobilization in a professional OKG-22 type sling.
Product’s description:
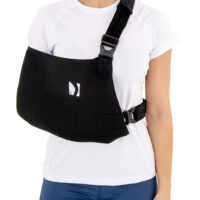
The OKG-22 Thumb -Loop left or right shoulder sling is a professional medical device that effectively immobilizes the upper limb during recovery from fractures, sprains or surgery.
Made of innovative breathable ActiveSpace™ material, it ensures constant air circulation, while the skin-friendly SoftComfort™ cotton padding enhances comfort.
The product has an innovative design, providing excellent comfort and support for both the left and right shoulder.
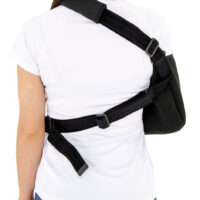
The circumferential strap prevents against involuntary arm movements, and the special thumb loop provides a comfortable position for the hand and prevents it from sagging.
Appropriately designed construction evenly distributes weight and supports the healing process.
A wide range of adjustment of the circumferential and shoulder strap allows individual size adjustment.
Soft cushion relieves pressure on bony prominences and prevents irritation of the skin of the neck area during use.
The thumb loop supports the hand and keeps the wrist in a neutral position for comfortable wear. It provides anatomical immobilization of the lower limb at 90 degrees at the elbow joint.
The OKG-22 orthopaedic thumb loop sling is a reliable support for people after upper limb injuries. It provides stabilization and pressure relief for the arm, elbow and shoulder, protecting against strain and aggravation of the injury.
Purpose of use:
– shoulder dislocation and sprain
– humerus, collarbone or elbow fractures
– after orthopedic surgeries of the shoulder and upper limb
– overusing and joint inflammation
Sizes
| Size | The length of the forearm and hand | How to measure |
| S | 33 – 41 cm | 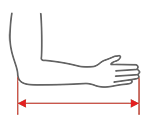 |
| M | 41 – 51cm | |
| L | 51 – 62 cm |
Fits for both forearms.
Total length of the product:
S:40 cm
M:45 cm
L: 50 cm
Gallery
Technology
MATERIALS
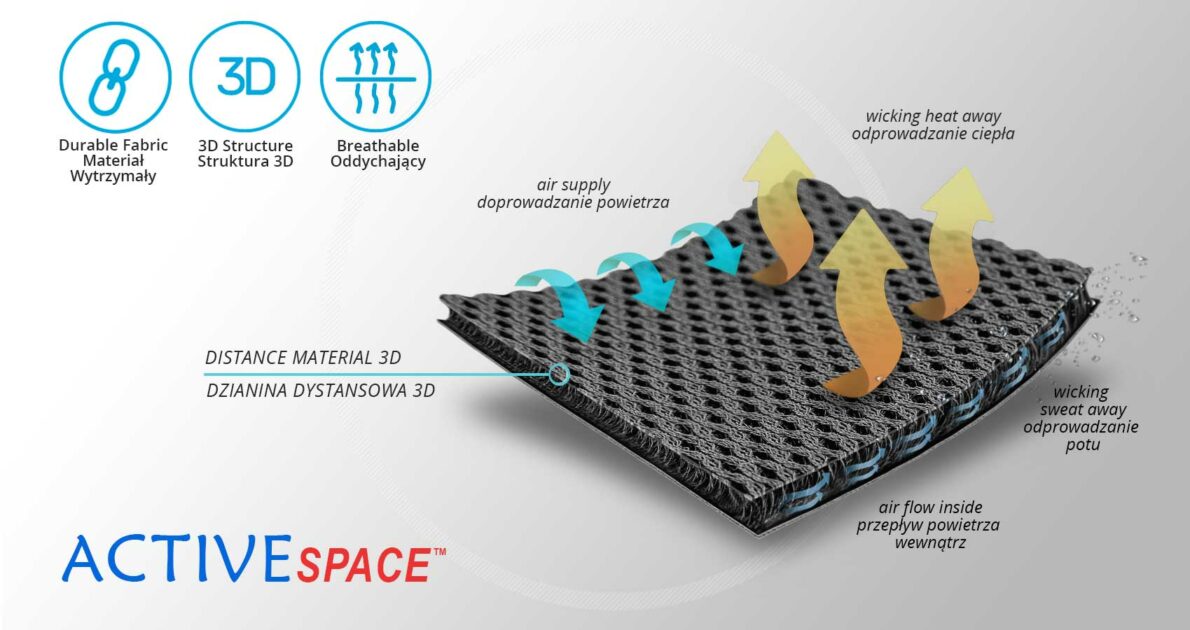
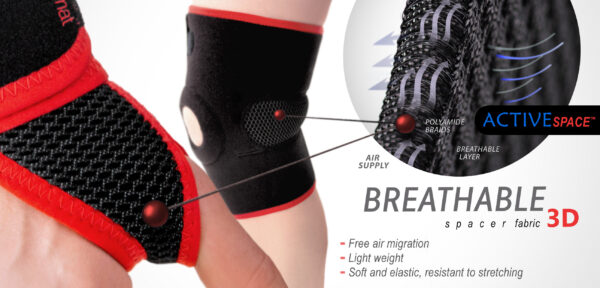
ActiveSpace™
ActiveSpace™ is a spacer, polyamide 3D lamination with high skin ventilation. It is very lightweight, consisted of 2 layers. Between them, we use polyamide braids. ActiveSpace™ is not elastic what improves stabilization. Inside the lamination, between 2 layers, the air flows freely, maintaining minimal water and moisture absorption. Waterproof material.
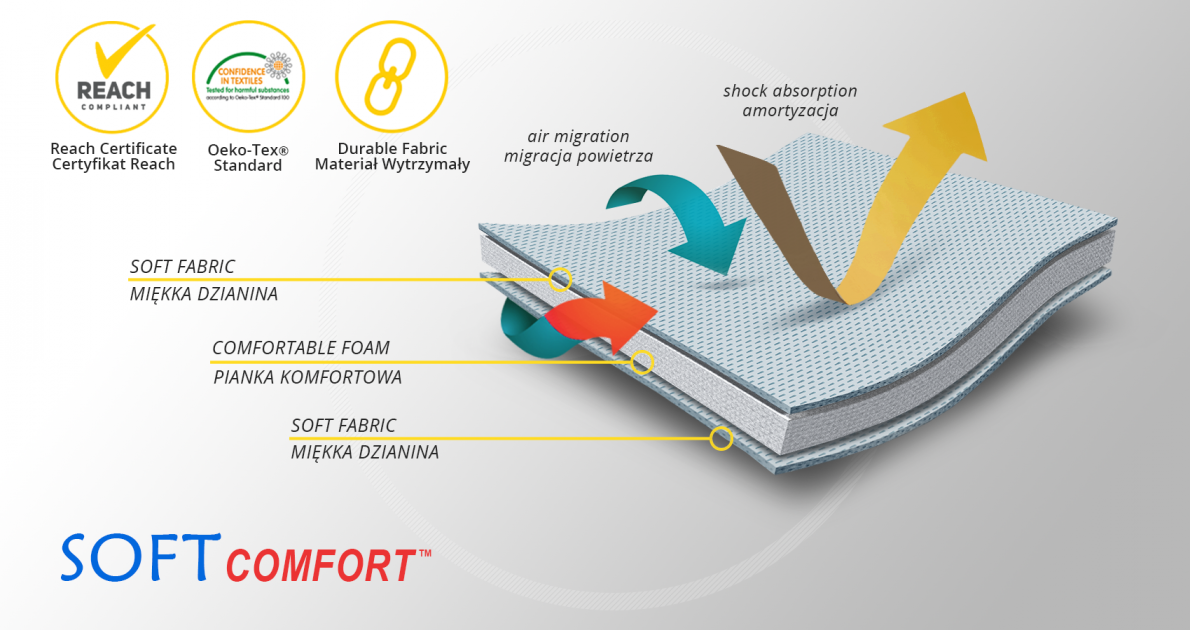
SoftComfort™
SoftComfort™ - a material specially designed to relieve very delicate places on the patient's body. The cover knit used in the production is made in such a way that it can come into contact with very sensitive skin. This raw material has the Oeko-Tex Standard 100 certificate and complies with the European REACH standard. The structure of the material consists of a very soft comfort foam laminated with a delicate cover knit. The fabric has a perforation that facilitates the flow of air through the material and allows the skin to breathe. The SoftComfort™ material is perfect for the production of linings relieving the rigid and inflexible elements of orthopedic orthoses.
STIFFENINGS
Double axis splint 2RP
Double axis splint 2RP is the orthopaedic splint that reflects anatomic knee movement while flexion. The drop locks do not change their position relative to the axis of a knee joint. 2RP splints are reinforced polycentric splints, equipped with an articulated mechanism, the main element of which are sliders moving on parabolic tracks. These type of splints is primarily used in knee braces, where strong lateral stabilization of the knee joint and precise regulation of its mobility are essential. The arms of the splint are made of high-quality aluminum alloy, hot-coated with a durable powder coating, what makes them very light. Moreover, their surface is resistant to patient’s sweat and salt contained in it. The drop locks are made of very durable and strong acid-resistant steel in which a special hole system with threads were made. The splints are waterproof and can be used in wet and humid environments.
The splint is equipped with the hyperextension knee lock and has following angles of joint flexion: free joint, 10, 30 ,40, 60, 75, 90 degrees and the following angles of joint extension: 10, 20, 30, 40 degrees. The ROM adjustment is made with a special, hardened screws placed into threaded holes marked with a specific angle value we want to lock. In this kind of splint, you can lock not only the angles of flexion and extension of a knee joint, but also the desired range of knee joint movement. Each brace equipped with 2RP splint comes with a free Allen key for these settings. This method of adjusting drop locks in orthopedic splints prevents from their setting by unauthorized persons who do not have a key. The bottom part of the drop lock has an oval shape that allows you to attach a soft anatomic pad made in 3D technology that separates drop lock from the knee. All movable elements of the splints move on galvanized steel rivets and in order to prevent them from seizing, special Teflon sliders were used in these places. Such a construction ensures their maintenance-free operation.
ATTENTION: If the brace is equipped with two side splints, the adjustment should always be made separately on each of the splints, setting the range of motion at the same angle of flexion, extension or lock. Failure to comply with this role will be resulted in the loss of the product warranty.

Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.