The bunion splint AM-OP-02

 Foot brace
Foot brace Class I medical device
Class I medical device Double-sided
Double-sided Innovative
Innovative Latex-free
Latex-free Skin-friendly
Skin-friendlyBUNION SPLINT
Description

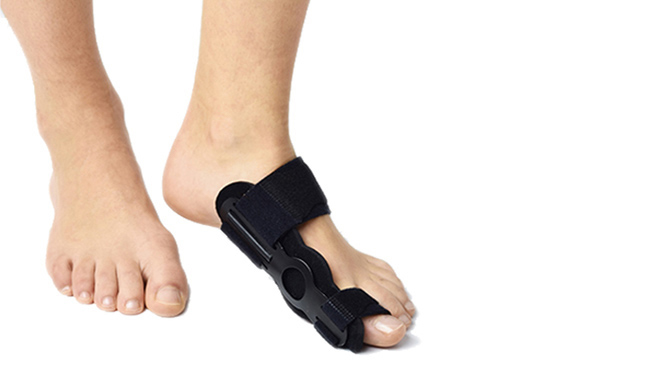
The bunion splint AM-OP-02 is intended for using as home and night supply. It corrects the hallux valgus and prevents against the metatarsus deformations.
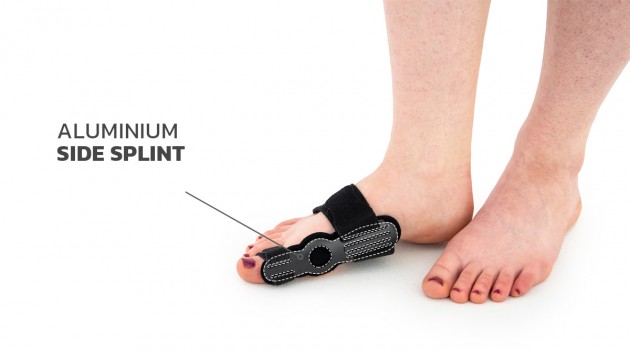
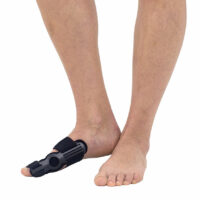
The splint is made of profiled lateral splint with hallux support, soft lining which is easy to attach, metatarsus straps and bunion strap. Special, soft lining soften the pressure and adjustable straps allow to fit device to individual foot’s shape and maintain it in required position. All you need is to fit the splint to the bunion and metatarsus with 2 circumferential straps. Wide metatarsus band nears the metatarsus bones to each other and improves the correction activity. Additionally, the splint is equipped with anatomic arch support, which lifts midfoot bones to providing plantar unloading. Friendly construction facilities putting the splint on and off, what makes it very easy and comfortable to use.
AM-OP-02 is intended for active correction of mild and medium hallux valgus. It achieves natural foot position and reduces bunion’s pain. The splint should be used as an prophylaxis and po-operative treatment. Thanks to splint the rehabilitation period of time is short and less uphill. You can use it both at home and during your sleep.
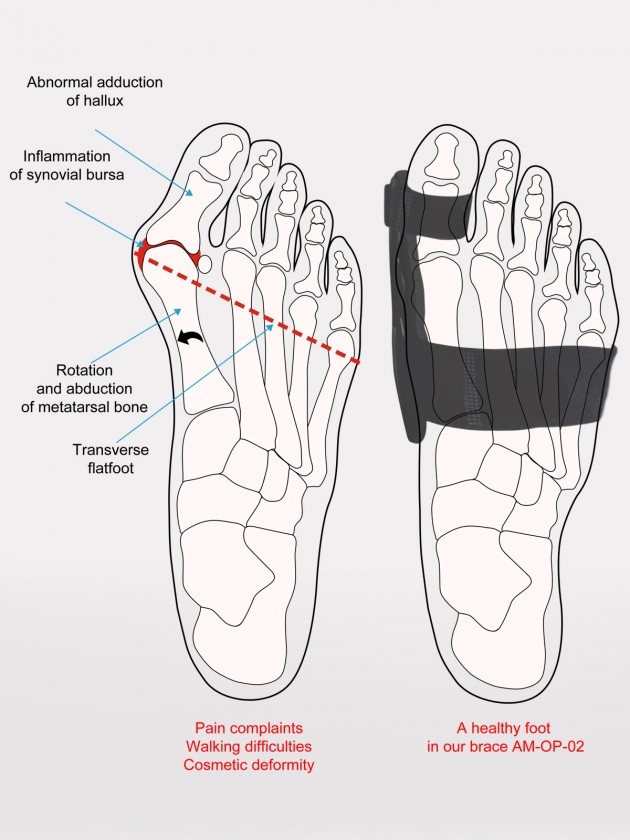
Our bunion splint arrests the hallux deformations, what can help to avoid the surgery. You can avoid the degenerative disease also as the result of decreasing the metatarsus joint inflammation risk.
Purpose of use
- Hallux rigidus
- degenerative disease in hallux metatarsophalangeal joint
- mild and medium bursitis
- postoperative treatment
Sizes
| Size | Metatarsal circumference | How to measure |
| M | 18 – 22 cm (7,1″ – 8,7″) |
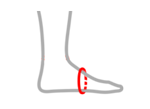 |
| L | 22 – 28 cm (8,9″ – 11″) |
Fits for both feet.
Total length: 14 cm (5,5″)
Gallery
Technology
MATERIALS
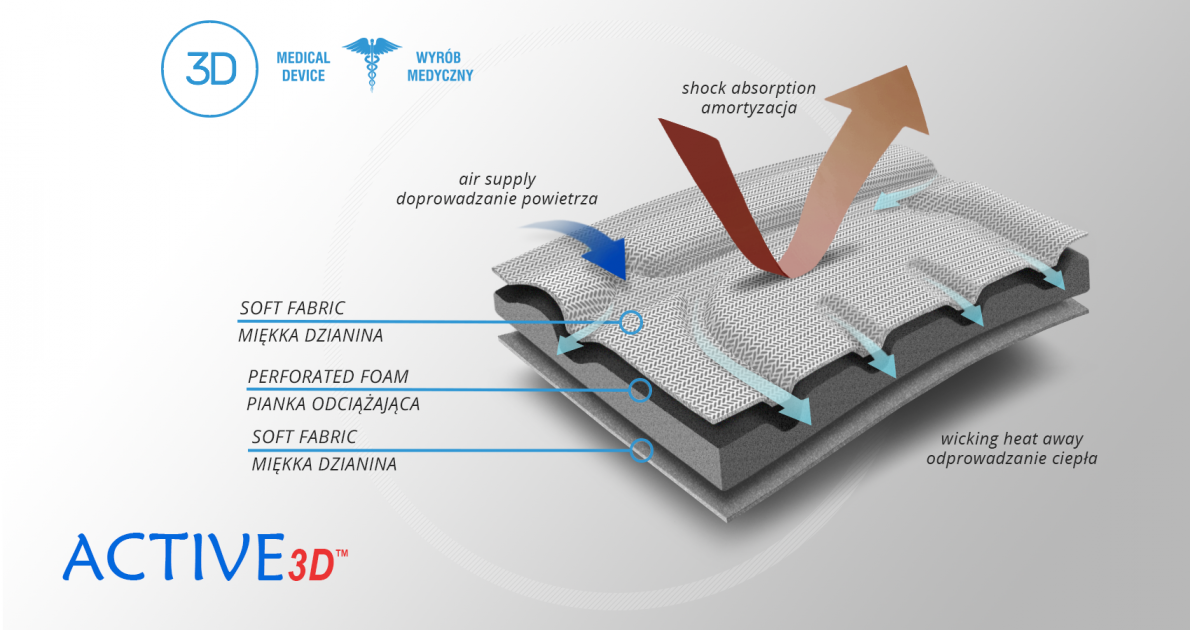
Active3D™
Active3D™ is thermoformed fabric made of special foamed, cell-closed designed material. It reduces the pressure on the body or any abrasions made by orthopaedic stays and aluminum splints. It is fully waterproof fabric and does not absorb sweat. It’s easy to clean. Due to its features, the fabric is an excellent product for making medical orthopaedic braces and orthoses. ACTIVE 3D™ has various external self-gripping layers. Our material has special, thermoformed properties and may be shaped according to the functional goals of the final braces.
UniTape™
UniTape™ is a technological orthopedic lamination with high durability with double-sided self-gripping function. UniTape™ is very thin and easy to attach what allows to fit the braces precisely.
STIFFENINGS
Bunion splint
PADDINGS
3D supports
3D relief supports are independent technical solutions to relieve the rigid elements of a given orthosis. These elements are made of supporting foams or EVA foam. These foams are connected with various types of skin-friendly materials and materials with an adhesive function. These pads have the appropriate shape and color adapted to the type of orthosis. They relieve both metal elements of orthoses, such as splints, stays, underwires and orthopedic drop locks, as well as other elements that should not come into direct contact with the patient's skin. These pads have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, guaranteeing the proper therapeutic effect.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.