Upper-extremity support OKG-13


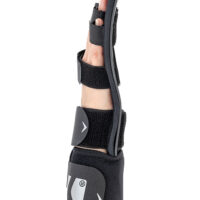
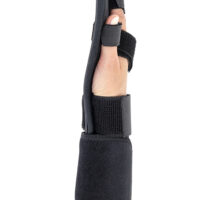
 Hand brace
Hand brace Wrist & forearm brace
Wrist & forearm brace Class I medical device
Class I medical device Compensation
Compensation Durable
Durable Neurology
Neurology Universal size
Universal size Waterproof
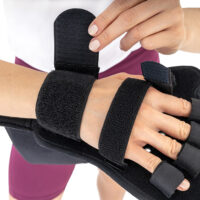
WaterproofWRIST ORTHOSIS WITH INDIVIDUAL FINGER SEPARATOR
Description
Made of lightweight, thermoformated ABS and covered with skin friendly fabric ActiveDistance™.
The soft element in the forearm part is made of AeroSpace II™.

Sizes
| Size | Wrist circumference | How to measure |
| Universal | min 11 – max 24 cm min 4,3″ – max 9,4″ |
 |
Right and left hand specific.
Total length of product: 36 cm (14,2″)
Gallery
Technology
MATERIALS
ActiveDistance™
ActiveDistance™ is an technical off-loading lamination with 3-layered construction. It’s made of EVA foam double-sided laminated with non-elastic polyamide layer that is easy to attach. This self-gripping function of ActiveDistance™ provides wide range of adjustment and perfect fitting. What is important, ActiveDistance™ is non-elastic fabric so it stabilizes your body in a perfect way. Thanks to using the comfort foam, the device is really soft what influences on the comfort of using it. Waterproof material.
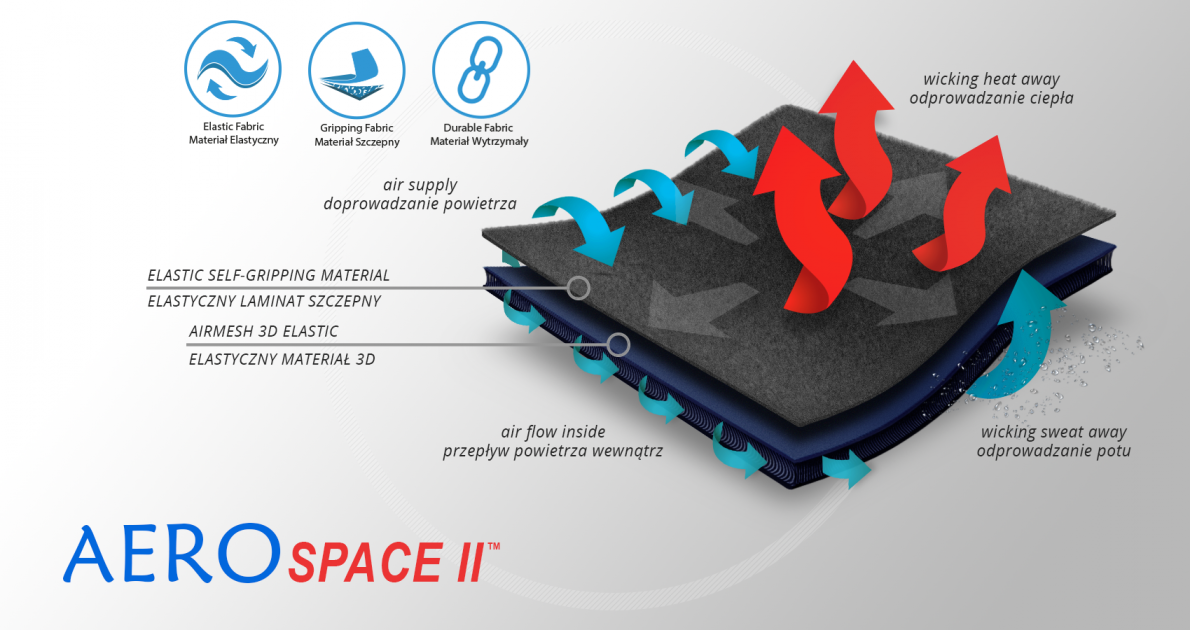
AeroSpace II™
AeroSpace II™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. AeroSpace II™ has an external self-gripping layer what makes it easy to adjust each product to individual patient’s needs. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.

STIFFENINGS
Flat aluminum stays
Aluminum stays are made of a special aluminum alloy that guarantees proper stiffness with minimal weight. They have rounded ends to prevent the damage of the product and come in various widths and thicknesses. The level of stabilization of the orthopedic device is defined by proper selection of the width and thickness of the aluminum stays. The stays can be pre-profiled or flat. They do not adapt to the shape of the patient's body, an individual adjustment of the orthosis is required by proper bending of the aluminum stays in the product. Thanks to this function, it is possible to correct the position of the patient's body or the secured joint.

Palm stiffening
Full stiffening of the palm brace is a unique solution made of ABS material. The stiffening is anatomically designed and covers the right and left hands. It has a system of special cutouts for elastic straps that stabilize each finger separately and the wrist. From the bottom of the patient’s palm there is a perforation system thanks to which the hand is ventilated.

Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.