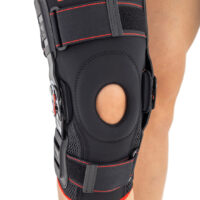
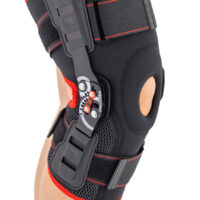
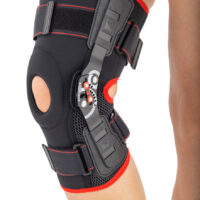
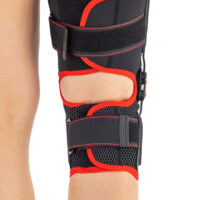
Knee support AS-KX-06
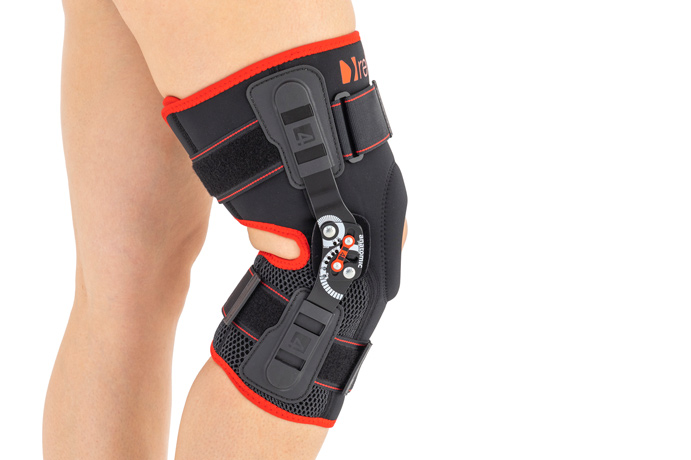

 Knee brace
Knee brace 2 in 1
2 in 1 Anatomic joint 2RA
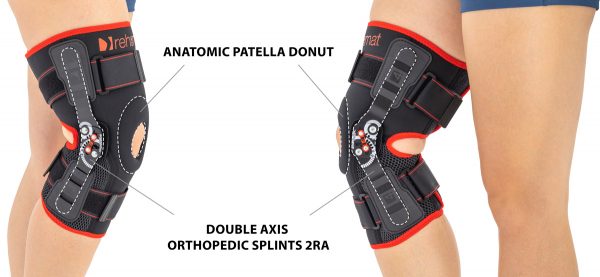
Anatomic joint 2RA Anatomic patella donut
Anatomic patella donut Breathable
Breathable Class I medical device
Class I medical device Latex-free
Latex-free Skin-friendly
Skin-friendlyREAR OPEN KNEE BRACE WITH ANATOMIC ROM ADJUSTMENT
Description
AS-KX-06 knee brace is made of fabric called ActivePren™. The parts are made of ActiveSpace™.
Our brace construction is unique because it has wrap around knee brace function (it’s easy to putting on) and closed brace function (very good patella and knee stabilization because of anatomic-shaped patella donut).
The brace is very good for drivers, because patient can sit into car seat, remove the brace very fast and easy and put it on before alighting from the car.
In addition, the brace has open popliteal fossa, what improves comfort of use and allows skin to breath.
AS-KX-06 knee brace is equipped with two pairs of circumferential straps and two sides 2RA precision splints that reflect anatomic knee motion.
Purpose of use
AS-KX-06 knee brace should be applied in cases of:
- knee dislocation,
- knee twist and sprain,
- side instability of the knee joint,
- knee ligaments LCL, MCL and ACL injuries
- knee ligaments reconstruction,
- other surgeries (orthopedic).
It is possible to use a thinner pad under the drop lock.
Sizes
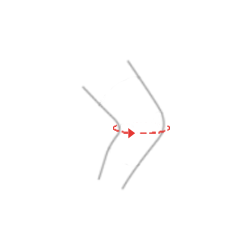
| Size | Knee circumference | How to measure |
| M | 34-38 cm (13,6″-15″) |
 |
| L | 38-42 cm (15,2″-16,5″) |
|
| XL | 42-46 cm (16,7″-18,1″) |
|
| XXL | 46-50 cm (18,3″-19,7″) |
Total length: 35 cm (13,8″)
Gallery
Technology
MATERIALS
ActivePren™
ActivePren™ is an active three-layer material consisting of two elastic jersey cover fabrics and a core made of neoprene foam. This material is characterized by softness and high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of ActivePren are the strongest and most effective stabilizing orthoses available on the market.
ActiveSpace™
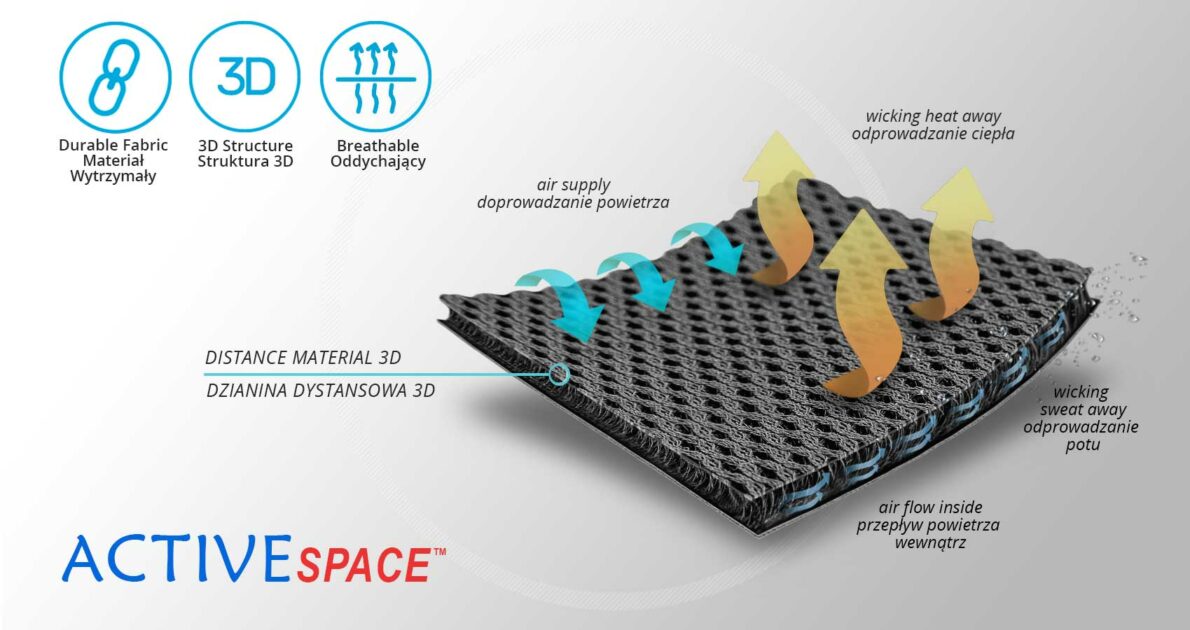
ActiveSpace™ is a spacer, polyamide 3D lamination with high skin ventilation. It is very lightweight, consisted of 2 layers. Between them, we use polyamide braids. ActiveSpace™ is not elastic what improves stabilization. Inside the lamination, between 2 layers, the air flows freely, maintaining minimal water and moisture absorption. Waterproof material.
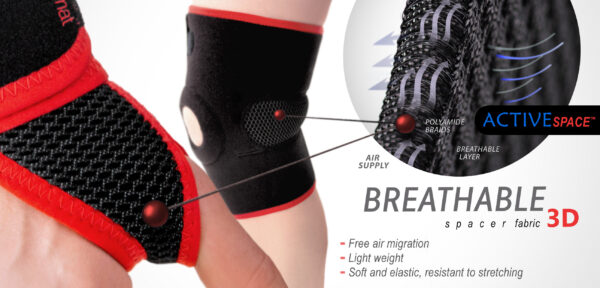
TECHNOLOGICAL SYSTEMS
2in1 SOLUTION - full comfort, simply use
 2in1 SOLUTION is another innovative solution that is aimed at perfect fitting of REH4MAT braces to different patients' needs. It is common knowledge that our main idea is to provide comfort and stabilization to the knee joint and ensure the most easy way of using our braces. That is why, work of our technologists has already ended with the real success! They have designed 2in1 SOLUTION and used it in some of our knee stabilizers.
2in1 SOLUTION is a unique project that connects both open knee brace function (it’s easy to putting on) and closed brace function (very good patella and knee stabilization). To ensure such a great stabilization the braces was equipped with anatomic-shaped patella donut). An additional advantage of 2in1 SOLUTION is an unrepeatable design - open popliteal fossa, what improves comfort of use. Such a solution allows skin for breathing and as a result the braces are wearing in a free and effortless way.
2in1 SOLUTION is another innovative solution that is aimed at perfect fitting of REH4MAT braces to different patients' needs. It is common knowledge that our main idea is to provide comfort and stabilization to the knee joint and ensure the most easy way of using our braces. That is why, work of our technologists has already ended with the real success! They have designed 2in1 SOLUTION and used it in some of our knee stabilizers.
2in1 SOLUTION is a unique project that connects both open knee brace function (it’s easy to putting on) and closed brace function (very good patella and knee stabilization). To ensure such a great stabilization the braces was equipped with anatomic-shaped patella donut). An additional advantage of 2in1 SOLUTION is an unrepeatable design - open popliteal fossa, what improves comfort of use. Such a solution allows skin for breathing and as a result the braces are wearing in a free and effortless way.
STIFFENINGS
Splint 2RA Precision
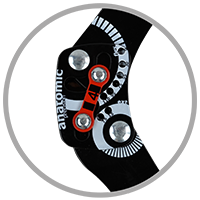 Splints 2RA Precision are a double axis anatomic splints which reflect the knee movement and during performance do not change their position towards the joint axis. Splints 2RA are double axis polycentric splints, with coupling mechanism with two main conductive elements which move of parabolic tracks. Such splints are mainly used in all knee joint braces and, apart from angle adjustment, the high lateral knee stabilization is very important. The splints are manufactured with high quality of sanded aluminium, durable cover which what makes splints neutral to the influence of sweat and salt. Flexion and extension angle adjustment is possible with special allen screws. Every brace is equipped with special Allen key which is necessary for adjustment. Anatomic splints provides hyperextension block, 30°, 55°, 75°, 95°, 110° flexion and 15° or 75° extension. The angle adjustment clock is secured from manipulation by unauthorized people. Standard length of the splints is: 290 and 390 mm. The splint and its design are patented in European Union by Reh4Mat company.
Splints 2RA Precision are a double axis anatomic splints which reflect the knee movement and during performance do not change their position towards the joint axis. Splints 2RA are double axis polycentric splints, with coupling mechanism with two main conductive elements which move of parabolic tracks. Such splints are mainly used in all knee joint braces and, apart from angle adjustment, the high lateral knee stabilization is very important. The splints are manufactured with high quality of sanded aluminium, durable cover which what makes splints neutral to the influence of sweat and salt. Flexion and extension angle adjustment is possible with special allen screws. Every brace is equipped with special Allen key which is necessary for adjustment. Anatomic splints provides hyperextension block, 30°, 55°, 75°, 95°, 110° flexion and 15° or 75° extension. The angle adjustment clock is secured from manipulation by unauthorized people. Standard length of the splints is: 290 and 390 mm. The splint and its design are patented in European Union by Reh4Mat company.
PADDINGS
3D supports
3D relief supports are independent technical solutions to relieve the rigid elements of a given orthosis. These elements are made of supporting foams or EVA foam. These foams are connected with various types of skin-friendly materials and materials with an adhesive function. These pads have the appropriate shape and color adapted to the type of orthosis. They relieve both metal elements of orthoses, such as splints, stays, underwires and orthopedic drop locks, as well as other elements that should not come into direct contact with the patient's skin. These pads have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, guaranteeing the proper therapeutic effect.
Patella stabilizers
Relief stabilizers of various shapes made in 2D technology. They are made of relieving foams connected on one side with a fabric friendly to the patient's skin, and on the other with a gripper, thanks to which the stabilizer can be attached to the adhesive element of the orthosis. Thanks to such a structure, these pads do not have to be sewn into the orthosis, and they can simply be fastened to it from the inside. These elements have an anatomical shape and are made of comfortable foam with proper hardness and elasticity, which guarantees the proper therapeutic effect.
Setting up
Downloads
Accessories
ACCESSORIES / PRODUCTS TO BE USED WITH
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.