Upper-extremity support AS-B


 Shoulder brace
Shoulder brace Breathable
Breathable Class I medical device
Class I medical device Compression
Compression Double-sided
Double-sided Latex-free
Latex-freeSHOULDER BRACE
Description
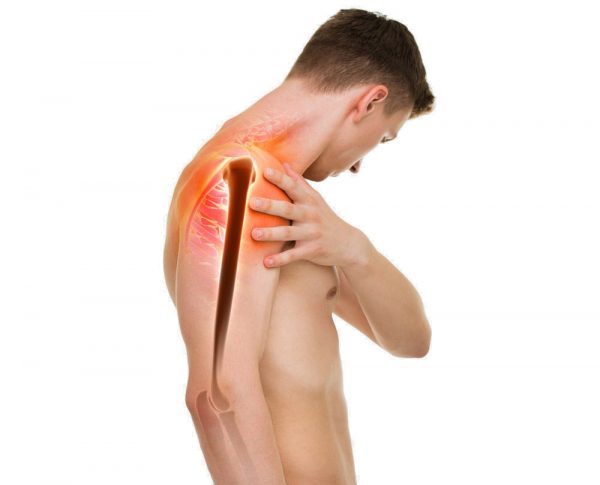
Shoulder instability
Shoulder instability can be an result of injury (e.g. dislocation) or other reasons (e.g. joint degeneration). The chronic instability, after shoulder dislocation, is very often. No matter what reason is, the instability is divided into: anterior, posterior, inferior and multidirectional instability. It can cause incarcerated rotator cuff tendons, cotyloid ligament compression, stretching of soft tissues and neve compression. It’s connected with pain, numbness and upper limb muscles weakness.
There’s a lot of sports predisposed to shoulder instability. There’re especially sports based on throwing (handball, water polo, baseball, basketball, volleyball), but contact sports also (rugby, American Football etc.).
Shoulder instability, however, must not mean end of the trainings. The fast and effective solution is our shoulder brace AS-B. The brace, providing the compression, supports the joint and prevents against the dislocations and contusions.
Product description
Our shoulder brace AS-B is an anatomic-shaped medical device is made of innovative fabrics called ActivePren™ and ActiveSpace™.
Shoulder brace AS-B with high-compressive properties, reduces effusions, swelling and pain. It prevents shoulder before and after injuries.
AS-B brace is the most effective tool to fight with shoulder instability for athletes.
Purpose of use:
– shoulder instability
– rotator cuff injury
– tendinopathy of shoulder
– prophylactically in sport
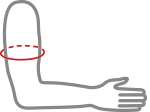
Sizes
| Size | Arm circumference | How to measure |
| S | 26-28 cm (10,4″-11″) |
 |
| M | 28-31 cm (11,2″-12,2″) |
|
| L | 31-34 cm (12,4″-13,4″) |
|
| XL | 34-36 cm (13,6″-14,2″) |
|
| XXL | 36-40 cm (14,4″-15,8″) |
|
| XXXL | 40-45 cm (16″-17,7″) |
Total length of the product: 31 cm (12,2″)
Gallery
Technology
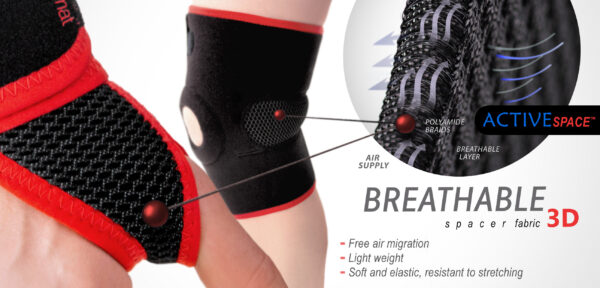
MATERIALS
ActivePren™
ActivePren™ is an active three-layer material consisting of two elastic jersey cover fabrics and a core made of neoprene foam. This material is characterized by softness and high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of ActivePren are the strongest and most effective stabilizing orthoses available on the market.
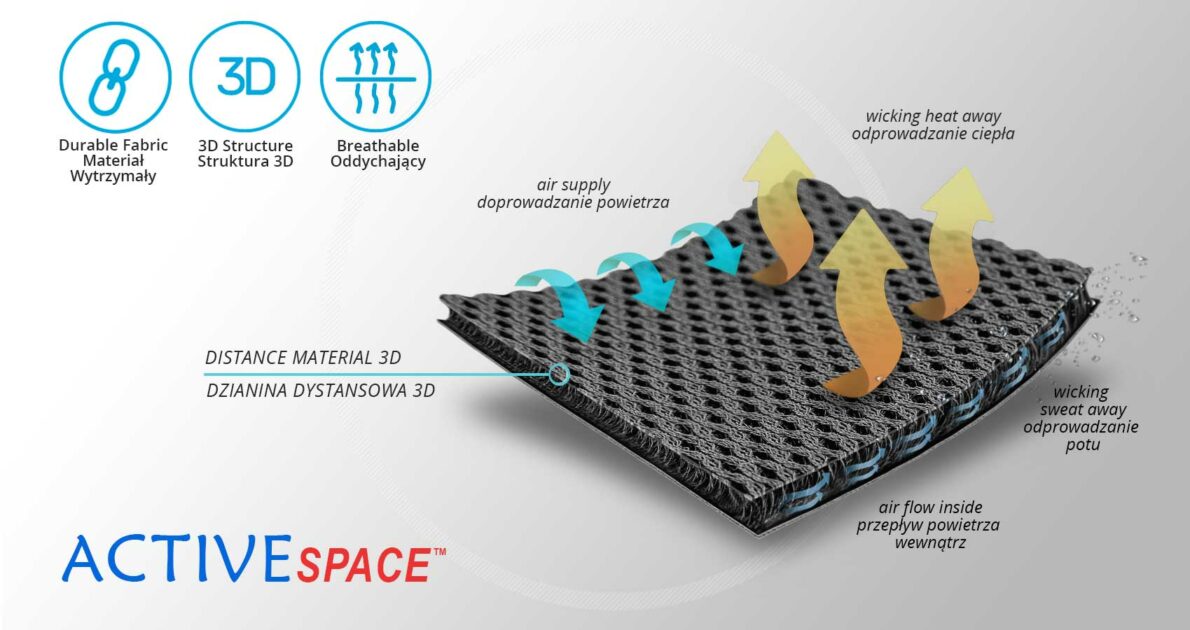
ActiveSpace™
ActiveSpace™ is a spacer, polyamide 3D lamination with high skin ventilation. It is very lightweight, consisted of 2 layers. Between them, we use polyamide braids. ActiveSpace™ is not elastic what improves stabilization. Inside the lamination, between 2 layers, the air flows freely, maintaining minimal water and moisture absorption. Waterproof material.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.