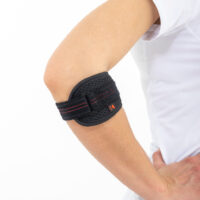
Elbow brace AM-LX-02


 Elbow brace
Elbow brace Tennis/golfer's elbow
Tennis/golfer's elbow Class I medical device
Class I medical device Double-sided
Double-sided Minimum device - maximum effect
Minimum device - maximum effect Silicone pad
Silicone padTennis elbow brace
Description
Tennis elbow develops as an result of lateral epicondyle inflammation in effect of forearm and wrist extensors and supinator muscle. It is called from tennis because it occurs in forehand movement but it’s possible in other sports (baseball, squash) and non-athlete activities.
Golf elbow is caused by micro-injuries of the medial epicondyle. A patient suffers from inflammation and pain, sometimes severe, on the inner part of the elbow. A lot of researchers claim that too tightly squeezing the stick causes stress of the forearm and wrist flexors that is why we call this condition Golfer’s elbow.
Innovative elbow strap AM-LX-02 with the silicone insert provides pressure on the afflicted muscles and tendons. Clinical trials show that over 80% of patients feel relief during using this kind of elbow brace.
The AM-LX-02 strap has an internal silicone insert that provides pressure on the muscles insertions and micro-massage during use of the support. Polyamide clip and VELCRO tape enable the adjustment of the strap to the limb.
Purpose of use
- Tennis elbow
- Golf elbow
Sizes
| Size | Forearm circumference | How to measure |
| Single size | min 22 cm – max 33 cm (min 8,7″ – max 13″) |
 |
Total height of the product: 6,5 cm (2,6″)
Gallery
Technology
MATERIALS
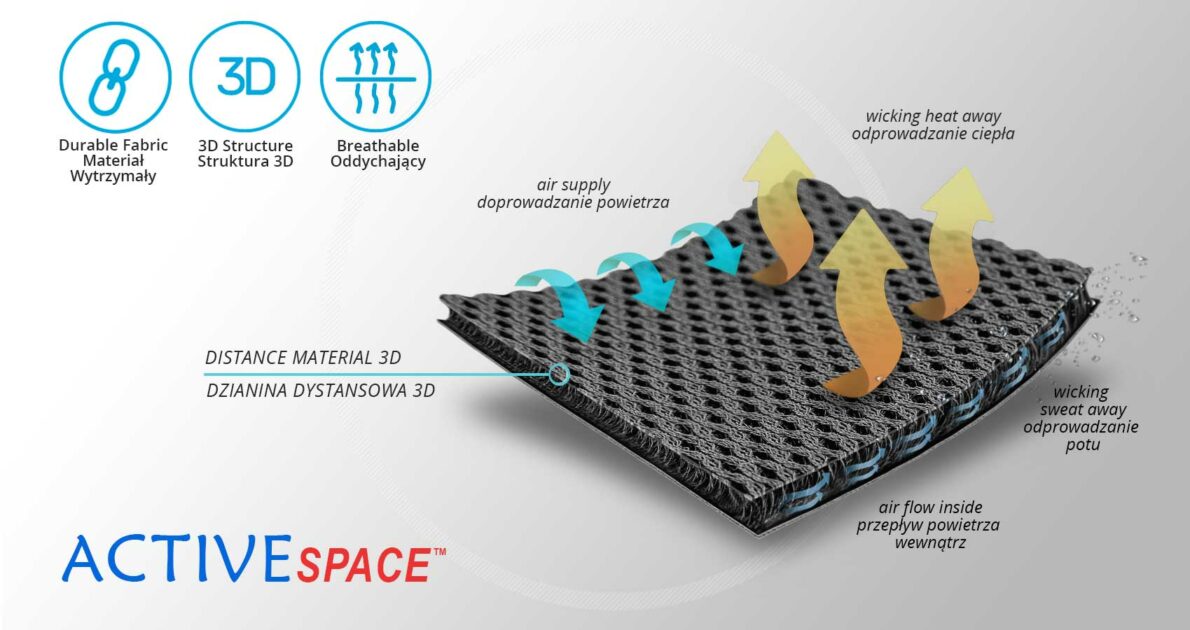
ActiveSpace™
ActiveSpace™ is a spacer, polyamide 3D lamination with high skin ventilation. It is very lightweight, consisted of 2 layers. Between them, we use polyamide braids. ActiveSpace™ is not elastic what improves stabilization. Inside the lamination, between 2 layers, the air flows freely, maintaining minimal water and moisture absorption. Waterproof material.
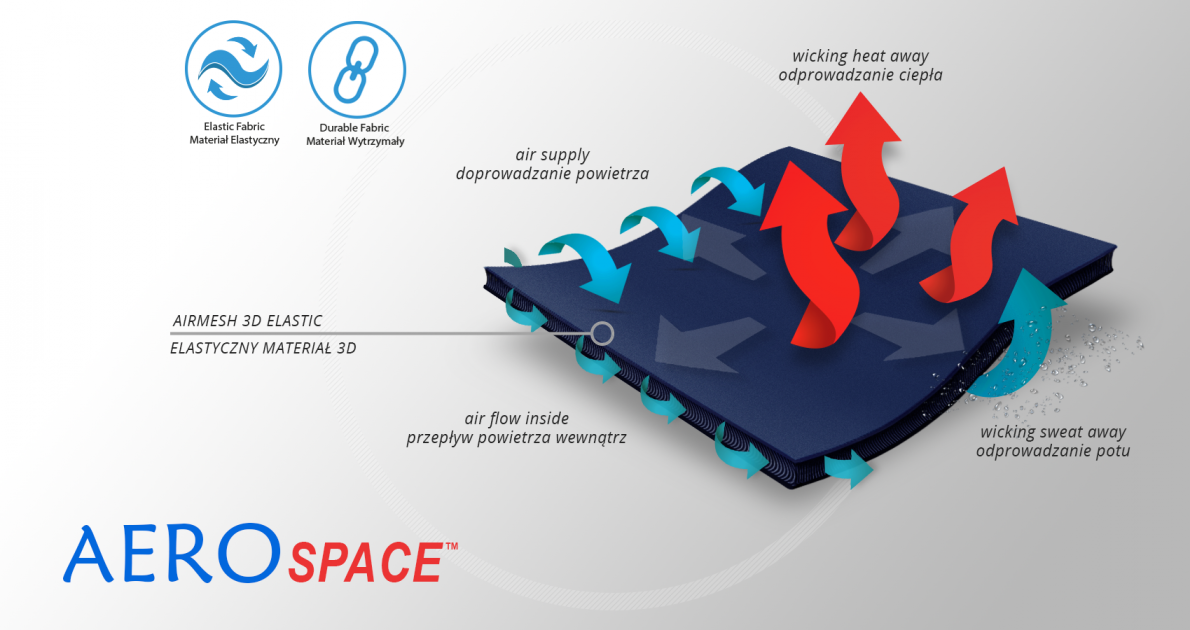
AeroSpace™
AeroSpace™ is an innovative new generation raw material. It is a distance elastic 3D knitted fabric consisting of two layers of facings and an interlacing that creates the appropriate thickness of the raw material and has relieving properties. The knitwear is made of the highest quality polyester yarn - guaranteeing high mechanical strength and spandex ensuring its proper flexibility. This material is characterized by a very low weight, high flexibility and a very large openwork structure, allowing for very easy drainage of sweat from the body and bringing fresh air to the skin. Products made of this raw material are neutral to the secured joint, do not heat or cool it, but ensure its proper compression and fit and reduce muscle vibrations generated during physical exertion. Its thickness and 3D structure perfectly relieves the orthopedic splints, stays or other elements mounted on the product and guarantees velvety softness to the touch.
TECHNOLOGICAL SYSTEMS
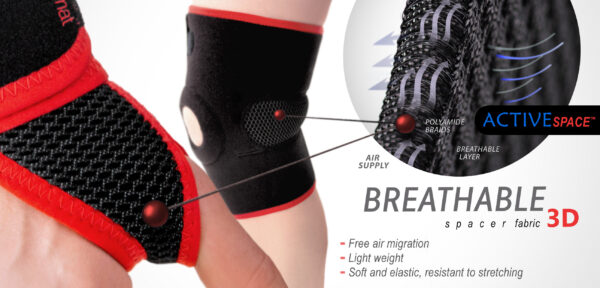
PROTECT BY SILICONE
![]()
PADDINGS
Tennis elbow oval silicone pads
Oval silicone pads with massage function used to eliminate the effects of tennis elbow injury. These pads have an anatomical shape and are made of silicone with the appropriate hardness and elasticity, guaranteeing the proper therapeutic effect.
Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.