Torso support OT-05


 LSO
LSO Class I medical device
Class I medical device Compression
Compression Durable
Durable Skin-friendly
Skin-friendly Waterproof
WaterproofOT-05 Industrial Work Back Brace with Suspender Straps
Description
CHRONIC LOWER BACK PAIN (CLBP)
Chronic lower back pain (CLBP) is the second leading cause of disability worldwide. Chronic lower back pain lasts for three months or longer. Sedentary lifestyles, overloading or operations requiring anaesthesia in the spine – these all cause back pain and make daily life difficult.
One of the causes of stressed spine and soft tissues such as muscles is heavy physical work, including non-ergonomic heavy lifting. The frequent shift of the body’s center of gravity (position), the heavy weight of the objects being lifted and the weakening of the so-called abdominal wall muscles, which are supposed to protect the spine from overload, cause excessive back strain and pain.
One of the proven preventive measures for a healthy back among industrial workers is the use of the professional pressure relieving back brace OT-05.
Product’s description:
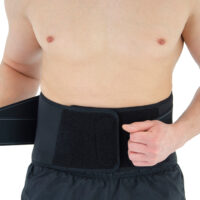
OT-05 back brace perfectly protects the lower back from strain during strenuous physical activities. The lower back brace consists of a professional elastic trunk belt with double-pull tension straps, lumbar stays and removable suspender straps.
The brace OT-05 is made of the innovative, highly breathable ActivePren™ material.
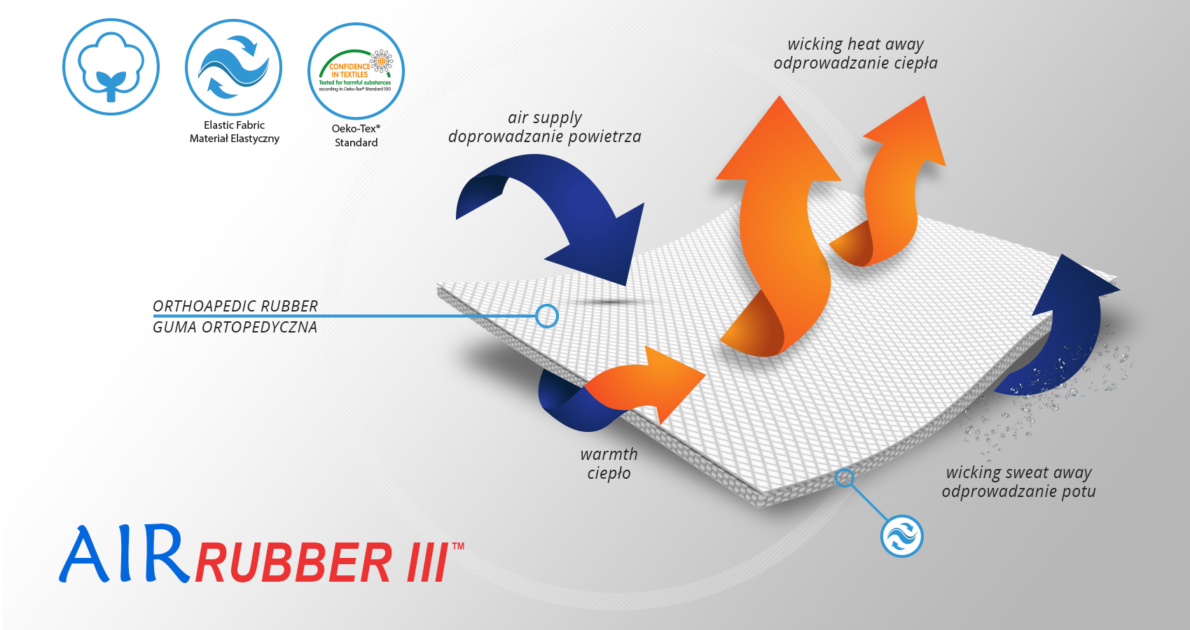
Double-pull tension straps are made of firm, elastic orthopaedic rubber AirRubber III™.
The trunk belt has been developed for industrial workers. The belt is fully breathable and absorbs the moisture from the skin. As a result, it can be used during physical work without any discomfort.

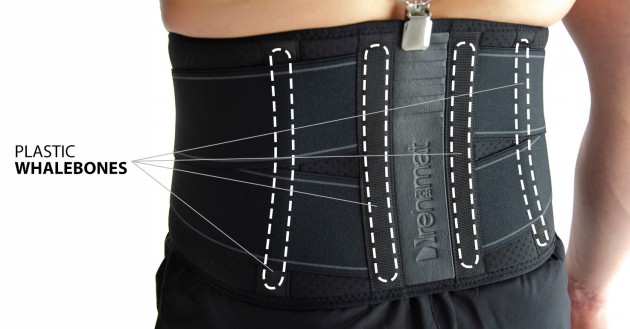
In the back of the belt, there are orthopaedic splints made of plastic that reduce the loading forces on the spine, thus relieving pressure on the intervertebral joints and spinal soft tissues (ligaments, muscles). Orthopaedic splints are lightweight and fully adapt to the physiological curvature of the spine, reducing back pain.
Unlike other heavy back braces, our OT-05 lower back industrial pain brace is made of breathable material that absorbs moisture whilst keeping your back well-supported.
High-strength hook-and-loop suspender straps holds the brace in place. It also offers a customized fit with the right amount of compression. Our high quality lumbar brace allows you to move around all day without your back brace falling off.

OT-05 Back Brace with Suspender Straps offers optimal support for lower back. Equipped with removable Y-shape suspenders to shape your body and provide compression when lifting heavy objects. Your back muscles, ligaments and vertebrae are protected against strain or sprain this way.
Our chronic pain lower back brace OT-05 is an excellent medical device for active people. Designed with lumbar stays for extra protection when you’re bending your back and helps soothe lower back pain, muscle spasms, and strains.
Our OT-05 Professional Work Back Brace with Suspender Straps offers wide range of adjustment and individual fit and compression. The unique desgin allows it to be used by both men and women. High-quality, durable materials and the optimal width of the product do not put pressure on the diaphragm and allow comfortable and safe ergonomic physical work.
Purpose of use:
- Pre- and Post-Surgical Stabilization,
- Degenerative Spinal pathologies,
- Disc Hernia,
- Spondylolithesis,
- Spondylolysis,
- Acute Back Pain,
- Spinal Instability,
- Rehabilitation and Prevention,
- herniated disc / discopathy,
- sciatica
Sizes
| Size | Waist circumference | How to measure |
|---|---|---|
| S | 65 – 75 cm (22,6″-29,5″) |
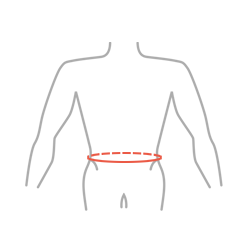 |
| M | 75 – 85 cm (29,5″-33,5″) |
|
| L | 85 – 97 cm (33,5″-38,2″) |
|
| XL | 97 – 110 cm (38,2″-43,3″) |
|
| 2XL | 110 – 125 cm (43,3″-49,2″) |
|
| 3XL | 125 – 145 cm (49,2″-57,1″) |
Total height of the product:
Front
S – 3XL: 15 cm
Back
S – 3XL: 20 cm
Gallery
Technology
MATERIALS
ActivePren™
ActivePren™ is an active three-layer material consisting of two elastic jersey cover fabrics and a core made of neoprene foam. This material is characterized by softness and high flexibility. A very important advantage of this material is the fact that it is not a knitted product, it does not have thick fibers, so that the weaves of the material do not imprint on the patient's skin and do not cause abrasionsin places of high compression. Products made of ActivePren are the strongest and most effective stabilizing orthoses available on the market.
AirRubber III™
AirRubber III™ has unidirectional elasticity. It can be stretched, increasing the length, not width, what improves compression. Between the braids air flows freely and skin can breathe. In addition in high compression, these rubber braids provide a gentle massage for skin. Our orthopedic rubber is very friendly for skin.
STIFFENINGS
Plastic stays
They come in various widths and thicknesses, are made of various types of plastics, such as polyamide, ABS or acrylic, and these features determine their stiffness. Thanks to their design, they are resistant to water, moisture and sweat. Products equipped with them can be washed without having to remove them from the orthosis. Our plastic stays work only in one direction, perfectly stabilize the laterally protected part of the body, adjusting to it at the same time and have a memory function, thanks to which they always return to their original shape. This function causes the stays in the orthosis to stabilize the swollen limb immediately after the injury and after the swelling has come off. The plastic stays cannot bend and that is why, they cannot correct the body posture or the secured joint.

Setting up
Downloads
ON OUR WEBSITE WE PRESENT MEDICAL DEVICES.
USE THEM ACCORDING TO THE INSTRUCTIONS FOR USE OR LABEL.

 Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
Class I medical device in accordance with Regulation (EU) 2017/745 of the European Parliament and of the Council of 5 April 2017. on medical devices.
MANUFACTURER / ADVERTISING ENTITY: REH4MAT Sp. z o.o.